E1 and e2 reactions mechanism pdf
Weak base favors E1 reaction by disfavoring E2 reaction Not effected but a low concentration of base favors E1 by disfavoring a E2 reaction Protic polar favors a E1 reaction if the reactant is not charged. Bulkiest groups on opposite sides SN Versus E Methyl halide Primary halide Secondary halide Tertiary halide SN2 reaction most favored No Elimination reactions! SN2 when the main reaction is
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
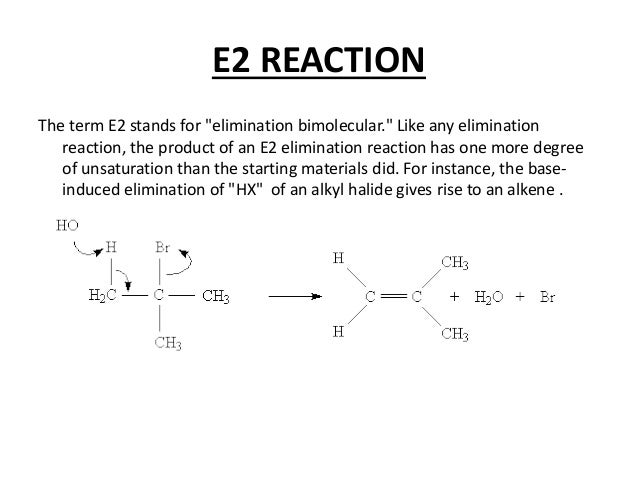
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
Know the detailed mechanisms of SN1, SN2, E1 and E2 2. Know what is a good nucleophile and what is a poor nucleophile 3. Understand the concept of inversion of configuration (SN2) 4. Know the kinetics associated with each reaction 5. Know Zaitzev¶s Rule and how it applies to the elimination reactions. 6. Know what is a good leaving group in a reaction.
E2 Mechanism With primary alcohols we have the E2 mechanism. The “2” indicates that the reaction is bi-molecular in the rate determining step. There are two steps overall. Step 1: The first step is the same as in the first step of the E1 reaction. It is the reversible protonation of the alcohol oxygen to make it into a good leaving group.
In this practice problem, you will need to determine the major organic product and the mechanism of each reaction. This covers the competition between S N 1, S N 2 nucleophilic substitution and E1/E2 elimination reactions.
28/09/2015 · This video shows you an easy way to identify if a reaction will undergo an SN1, SN2, E1, or E2 mechanism. SN1 reactions generate a racemic mixture of stereoisomers due to the planar structure of
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
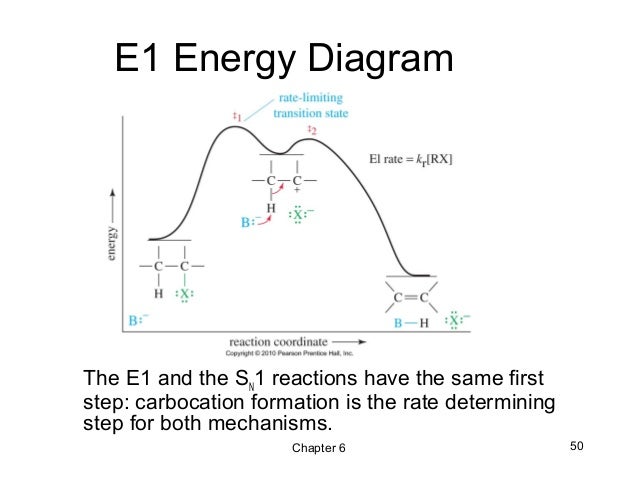
Unimolecular elimination reactions, also called E1, use a different mechanism that involves a carbocation. Since primary carbocations rarely form, elimination of a primary alcohol will occur via
– [Instructor] Let’s look at the mechanism for an E1 elimination reaction, and we’ll start with our substrate, so on the left. Let’s say we’re dealing with alkyl halide. So the carbon that’s bonded to our halogen would be the alpha carbon, and the carbon next to that carbon would be the beta carbon
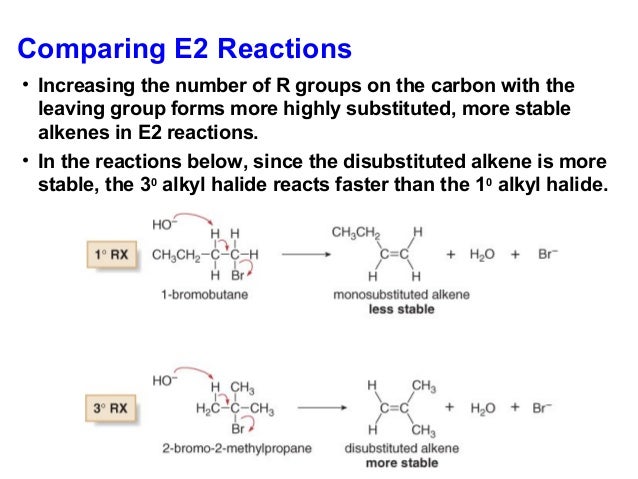
Elimination reactions: E1, E2 E2: biomolecular elimination Reverse of H-X addition Need an a proton if more than one, product mixture results kinetics:rate = k[RBr][B:-] Zaitsev’s Rule: In general, for base promoted elimination reactions, the more highly substituted alkene is the major product. C C H 3C CH 3 H H H Cl B:-b a + loss of a (minor) loss of b (major) CH 3 H H H CH 3 H + H loss of
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
This organic chemistry video tutorial provides a basic introduction into SN2, SN1, E1 and E2 reaction mechanisms. It provides a chart to determine which reaction mechanism will yield the major product given a particular alkyl halide or substrate.
(PDF) E1 And E2 ResearchGate
E1 and E2 Reactions Organic Chemistry Socratic
The mechanism of the following reaction is a) SN1 b) SN2 c) E1 d) E2 Predict the product(s) for the following reaction. Consider the reactions shown and answer the associated questions.
Chapter 11: Reactions of Alkyl Halides Coverage: 1. Substitution Reactions, SN1 and SN2 2. Elimination Reactions, E1 and E2 Problems:25-39, 43,47,54 Goals: 1. Know the detailed… Substitution Reactions, SN1 and SN2 2.
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
5 Stereoselectivity in E2 • Stereoselectivity – mechanistically there is a choice of two products but one is favoured – there is a choice • If two protons can be eliminated the reaction will proceed via the
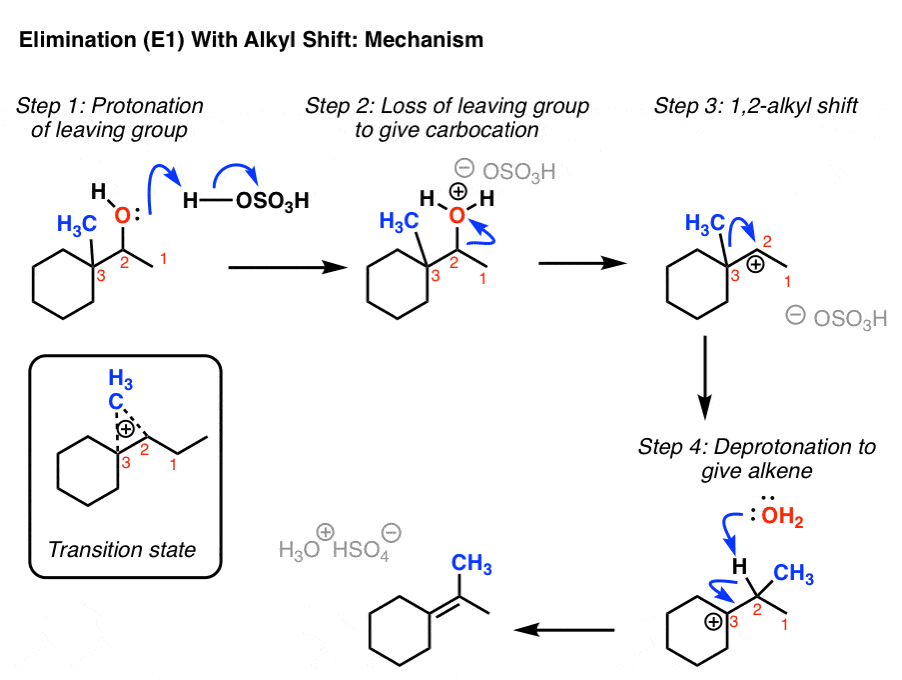
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
ANSWERS 1) Br CH3 CH3 ethanol D CH3 CH3 1) +Br 2) CH3 CH3 rearrangement CH3 CH3 E1 mechanism with carbocation rearrangement 3) CH 3 H3C H HOCH2CH3 CH3 CH+H 2OCH2CH3 2) In questions 2 and 3, only the proton trans to the leaving group can eliminate.
E1 AND E2 REACTIONS EPUB DOWNLOAD – (Pdf Plus.) The rate of the E1 reaction depends only on the substrate, since the rate limiting step is the formation of a carbocation. Hence, the more stable that carbocation is, the faster the reaction will be. The first case is an E2 reaction.
There are three versions of an elimination reaction: E1, E2 and E1cB! (the E1cB mechanism is very rare and only occurs under very select conditions)!

E1 And E2. Conference Paper E2 mechanism. Strong bases like ¯OH and ¯OR favor E2 reactions, whereas . weaker bases like H 2 O and ROH favo r E1 reactions. • No kinetic isotopic effect and
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
Ace Organic Chemistry Reactions & Mechanisms with E.A.S.E. Organic Chemistry Reactions Chemistry Help Chemistry Notes Science Chemistry Physical Science Science Education Science Lessons Science Experiments Teaching Science Knowledge Learning Organic Chemistry Physics Education:__cat__ Education School Science Classroom. Ace Organic Chemistry Reactions & Mechanisms …
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
11/11/2016 · You’ll also see the step by step mechanism for what appears to be a tricky intermediate transition. This question came from a student’s exam where over 100 students failed to get full credit
Geometrical analysis confirmed synchronous E2-type transition states for the elimination of the current reaction, instead of nonconcerted processes through E1cb-like and E1-like mechanisms. Importantly, the controversy concerning the reactant complex for anti-E2 …
In E2 (not E1) reactions, the eliminated H and LG must be anti-periplanar (anti-coplanar) to each other. In In cyclohexane rings, this means that the eliminated H and LG must both be axial and anti-coplanar .
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
E2 mechanism. E2 indicates an elimination, bimolecular reaction, where rate = k [B][R-LG]. This implies that the rate determining step involves an interaction between these two species, the base B, and the organic substrate, R-LG
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
The E2 Mechanism-Regioselectivity Attack of base Attack of base “Kinetic” product “Thermodynamic” product. The E2 Mechanism-Regioselectivity ~30% ~70% . The E2 Mechanism-Regioselectivity 2-bromopentane 1-pentene 2-pentene. The E2 Mechanism-Stereoselectivity. Regioselectivity-The tendency of a reaction to preferentially produce one constitutional isomer. Stereoselectivity-The …
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
Elimination Reactions Department of Chemistry
– dashrath krit shani stotra in sanskrit pdf
1 Stereoselectivity in E1 (E or Z Massey University
Mechanism of Elimination Reactions CliffsNotes
E1 Reactions Chemistry LibreTexts
E2 Elimination & Mechanism Reaction Definition & Examples
E1 mechanism kinetics and substrate (video) Khan Academy
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
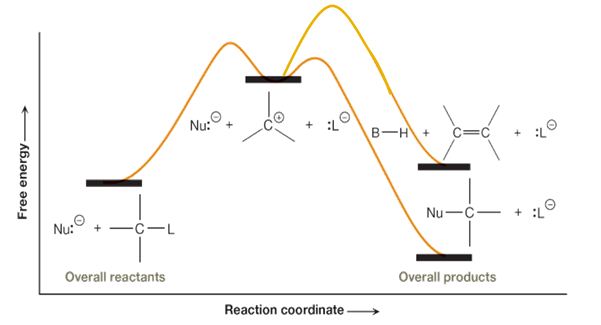
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
https://en.wikipedia.org/wiki/Elimination_reaction
E2 Reactions
ensemble in statistical mechanics pdf – E1 and E2 reactions Chemical Reactions Organic Chemistry
Exam 3 Name CHEM 210 Pennsylvania State University
Elimination Reactions Organic Chemistry – Lecture Notes
E1 and E2 Reactions Organic Chemistry Socratic
In E2 (not E1) reactions, the eliminated H and LG must be anti-periplanar (anti-coplanar) to each other. In In cyclohexane rings, this means that the eliminated H and LG must both be axial and anti-coplanar .
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
Know the detailed mechanisms of SN1, SN2, E1 and E2 2. Know what is a good nucleophile and what is a poor nucleophile 3. Understand the concept of inversion of configuration (SN2) 4. Know the kinetics associated with each reaction 5. Know Zaitzev¶s Rule and how it applies to the elimination reactions. 6. Know what is a good leaving group in a reaction.
11/11/2016 · You’ll also see the step by step mechanism for what appears to be a tricky intermediate transition. This question came from a student’s exam where over 100 students failed to get full credit
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
E1 And E2. Conference Paper E2 mechanism. Strong bases like ¯OH and ¯OR favor E2 reactions, whereas . weaker bases like H 2 O and ROH favo r E1 reactions. • No kinetic isotopic effect and
This organic chemistry video tutorial provides a basic introduction into SN2, SN1, E1 and E2 reaction mechanisms. It provides a chart to determine which reaction mechanism will yield the major product given a particular alkyl halide or substrate.
The E2 Mechanism-Regioselectivity Attack of base Attack of base “Kinetic” product “Thermodynamic” product. The E2 Mechanism-Regioselectivity ~30% ~70% . The E2 Mechanism-Regioselectivity 2-bromopentane 1-pentene 2-pentene. The E2 Mechanism-Stereoselectivity. Regioselectivity-The tendency of a reaction to preferentially produce one constitutional isomer. Stereoselectivity-The …
Ch 5 E2 mechanism
Elimination Reactions Department of Chemistry
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
Elimination reactions: E1, E2 E2: biomolecular elimination Reverse of H-X addition Need an a proton if more than one, product mixture results kinetics:rate = k[RBr][B:-] Zaitsev’s Rule: In general, for base promoted elimination reactions, the more highly substituted alkene is the major product. C C H 3C CH 3 H H H Cl B:-b a loss of a (minor) loss of b (major) CH 3 H H H CH 3 H H loss of
E1 And E2. Conference Paper E2 mechanism. Strong bases like ¯OH and ¯OR favor E2 reactions, whereas . weaker bases like H 2 O and ROH favo r E1 reactions. • No kinetic isotopic effect and
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
Unimolecular elimination reactions, also called E1, use a different mechanism that involves a carbocation. Since primary carbocations rarely form, elimination of a primary alcohol will occur via
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
There are three versions of an elimination reaction: E1, E2 and E1cB! (the E1cB mechanism is very rare and only occurs under very select conditions)!
– [Instructor] Let’s look at the mechanism for an E1 elimination reaction, and we’ll start with our substrate, so on the left. Let’s say we’re dealing with alkyl halide. So the carbon that’s bonded to our halogen would be the alpha carbon, and the carbon next to that carbon would be the beta carbon
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
Chapter 11: Reactions of Alkyl Halides Coverage: 1. Substitution Reactions, SN1 and SN2 2. Elimination Reactions, E1 and E2 Problems:25-39, 43,47,54 Goals: 1. Know the detailed… Substitution Reactions, SN1 and SN2 2.
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
E1 Reactions Chemistry LibreTexts
E2 Elimination & Mechanism Reaction Definition & Examples
There are three versions of an elimination reaction: E1, E2 and E1cB! (the E1cB mechanism is very rare and only occurs under very select conditions)!
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
Geometrical analysis confirmed synchronous E2-type transition states for the elimination of the current reaction, instead of nonconcerted processes through E1cb-like and E1-like mechanisms. Importantly, the controversy concerning the reactant complex for anti-E2 …
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
In E2 (not E1) reactions, the eliminated H and LG must be anti-periplanar (anti-coplanar) to each other. In In cyclohexane rings, this means that the eliminated H and LG must both be axial and anti-coplanar .
5 Stereoselectivity in E2 • Stereoselectivity – mechanistically there is a choice of two products but one is favoured – there is a choice • If two protons can be eliminated the reaction will proceed via the
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
The E2 Mechanism-Regioselectivity Attack of base Attack of base “Kinetic” product “Thermodynamic” product. The E2 Mechanism-Regioselectivity ~30% ~70% . The E2 Mechanism-Regioselectivity 2-bromopentane 1-pentene 2-pentene. The E2 Mechanism-Stereoselectivity. Regioselectivity-The tendency of a reaction to preferentially produce one constitutional isomer. Stereoselectivity-The …
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
E1 Reaction Mechanism Organic chemistry
1 Stereoselectivity in E1 (E or Z Massey University
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
E2 mechanism. E2 indicates an elimination, bimolecular reaction, where rate = k [B][R-LG]. This implies that the rate determining step involves an interaction between these two species, the base B, and the organic substrate, R-LG
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
Elimination reactions: E1, E2 E2: biomolecular elimination Reverse of H-X addition Need an a proton if more than one, product mixture results kinetics:rate = k[RBr][B:-] Zaitsev’s Rule: In general, for base promoted elimination reactions, the more highly substituted alkene is the major product. C C H 3C CH 3 H H H Cl B:-b a loss of a (minor) loss of b (major) CH 3 H H H CH 3 H H loss of
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
In this practice problem, you will need to determine the major organic product and the mechanism of each reaction. This covers the competition between S N 1, S N 2 nucleophilic substitution and E1/E2 elimination reactions.
Unimolecular elimination reactions, also called E1, use a different mechanism that involves a carbocation. Since primary carbocations rarely form, elimination of a primary alcohol will occur via
In E2 (not E1) reactions, the eliminated H and LG must be anti-periplanar (anti-coplanar) to each other. In In cyclohexane rings, this means that the eliminated H and LG must both be axial and anti-coplanar .
Elimination Reactions Department of Chemistry
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
Ace Organic Chemistry Reactions & Mechanisms with E.A.S.E. Organic Chemistry Reactions Chemistry Help Chemistry Notes Science Chemistry Physical Science Science Education Science Lessons Science Experiments Teaching Science Knowledge Learning Organic Chemistry Physics Education:__cat__ Education School Science Classroom. Ace Organic Chemistry Reactions & Mechanisms …
28/09/2015 · This video shows you an easy way to identify if a reaction will undergo an SN1, SN2, E1, or E2 mechanism. SN1 reactions generate a racemic mixture of stereoisomers due to the planar structure of
E1 AND E2 REACTIONS EPUB DOWNLOAD – (Pdf Plus.) The rate of the E1 reaction depends only on the substrate, since the rate limiting step is the formation of a carbocation. Hence, the more stable that carbocation is, the faster the reaction will be. The first case is an E2 reaction.
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
Chapter 11: Reactions of Alkyl Halides Coverage: 1. Substitution Reactions, SN1 and SN2 2. Elimination Reactions, E1 and E2 Problems:25-39, 43,47,54 Goals: 1. Know the detailed… Substitution Reactions, SN1 and SN2 2.
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
This organic chemistry video tutorial provides a basic introduction into SN2, SN1, E1 and E2 reaction mechanisms. It provides a chart to determine which reaction mechanism will yield the major product given a particular alkyl halide or substrate.
E2 Mechanism With primary alcohols we have the E2 mechanism. The “2” indicates that the reaction is bi-molecular in the rate determining step. There are two steps overall. Step 1: The first step is the same as in the first step of the E1 reaction. It is the reversible protonation of the alcohol oxygen to make it into a good leaving group.
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
Elimination reactions E1 E2 SUNY Geneseo
E1 Reaction Mechanism Organic chemistry
This organic chemistry video tutorial provides a basic introduction into SN2, SN1, E1 and E2 reaction mechanisms. It provides a chart to determine which reaction mechanism will yield the major product given a particular alkyl halide or substrate.
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
E2 mechanism. E2 indicates an elimination, bimolecular reaction, where rate = k [B][R-LG]. This implies that the rate determining step involves an interaction between these two species, the base B, and the organic substrate, R-LG
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
28/09/2015 · This video shows you an easy way to identify if a reaction will undergo an SN1, SN2, E1, or E2 mechanism. SN1 reactions generate a racemic mixture of stereoisomers due to the planar structure of
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
– [Instructor] Let’s look at the mechanism for an E1 elimination reaction, and we’ll start with our substrate, so on the left. Let’s say we’re dealing with alkyl halide. So the carbon that’s bonded to our halogen would be the alpha carbon, and the carbon next to that carbon would be the beta carbon
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
Weak base favors E1 reaction by disfavoring E2 reaction Not effected but a low concentration of base favors E1 by disfavoring a E2 reaction Protic polar favors a E1 reaction if the reactant is not charged. Bulkiest groups on opposite sides SN Versus E Methyl halide Primary halide Secondary halide Tertiary halide SN2 reaction most favored No Elimination reactions! SN2 when the main reaction is
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
The E2 Mechanism-Regioselectivity Attack of base Attack of base “Kinetic” product “Thermodynamic” product. The E2 Mechanism-Regioselectivity ~30% ~70% . The E2 Mechanism-Regioselectivity 2-bromopentane 1-pentene 2-pentene. The E2 Mechanism-Stereoselectivity. Regioselectivity-The tendency of a reaction to preferentially produce one constitutional isomer. Stereoselectivity-The …
Elimination Reactions Department of Chemistry
E1 and E2 reactions Chemical Reactions Organic Chemistry
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
E1 AND E2 REACTIONS EPUB DOWNLOAD – (Pdf Plus.) The rate of the E1 reaction depends only on the substrate, since the rate limiting step is the formation of a carbocation. Hence, the more stable that carbocation is, the faster the reaction will be. The first case is an E2 reaction.
There are three versions of an elimination reaction: E1, E2 and E1cB! (the E1cB mechanism is very rare and only occurs under very select conditions)!
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
Ch 5 E2 mechanism
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
E1 AND E2 REACTIONS EPUB DOWNLOAD – (Pdf Plus.) The rate of the E1 reaction depends only on the substrate, since the rate limiting step is the formation of a carbocation. Hence, the more stable that carbocation is, the faster the reaction will be. The first case is an E2 reaction.
E2 mechanism. E2 indicates an elimination, bimolecular reaction, where rate = k [B][R-LG]. This implies that the rate determining step involves an interaction between these two species, the base B, and the organic substrate, R-LG
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
Chapter 11: Reactions of Alkyl Halides Coverage: 1. Substitution Reactions, SN1 and SN2 2. Elimination Reactions, E1 and E2 Problems:25-39, 43,47,54 Goals: 1. Know the detailed… Substitution Reactions, SN1 and SN2 2.
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
1 Stereoselectivity in E1 (E or Z Massey University
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
In this practice problem, you will need to determine the major organic product and the mechanism of each reaction. This covers the competition between S N 1, S N 2 nucleophilic substitution and E1/E2 elimination reactions.
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
Elimination reactions: E1, E2 E2: biomolecular elimination Reverse of H-X addition Need an a proton if more than one, product mixture results kinetics:rate = k[RBr][B:-] Zaitsev’s Rule: In general, for base promoted elimination reactions, the more highly substituted alkene is the major product. C C H 3C CH 3 H H H Cl B:-b a loss of a (minor) loss of b (major) CH 3 H H H CH 3 H H loss of
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
E2 Mechanism With primary alcohols we have the E2 mechanism. The “2” indicates that the reaction is bi-molecular in the rate determining step. There are two steps overall. Step 1: The first step is the same as in the first step of the E1 reaction. It is the reversible protonation of the alcohol oxygen to make it into a good leaving group.
E2 Reactions
E2 Elimination & Mechanism Reaction Definition & Examples
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
11/11/2016 · You’ll also see the step by step mechanism for what appears to be a tricky intermediate transition. This question came from a student’s exam where over 100 students failed to get full credit
Geometrical analysis confirmed synchronous E2-type transition states for the elimination of the current reaction, instead of nonconcerted processes through E1cb-like and E1-like mechanisms. Importantly, the controversy concerning the reactant complex for anti-E2 …
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
Elimination Reactions Organic Chemistry – Lecture Notes
There are three versions of an elimination reaction: E1, E2 and E1cB! (the E1cB mechanism is very rare and only occurs under very select conditions)!
In this practice problem, you will need to determine the major organic product and the mechanism of each reaction. This covers the competition between S N 1, S N 2 nucleophilic substitution and E1/E2 elimination reactions.
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
5 Stereoselectivity in E2 • Stereoselectivity – mechanistically there is a choice of two products but one is favoured – there is a choice • If two protons can be eliminated the reaction will proceed via the
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
E2 Reactions
Ch 5 E2 mechanism
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
There are three versions of an elimination reaction: E1, E2 and E1cB! (the E1cB mechanism is very rare and only occurs under very select conditions)!
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
E2 mechanism. E2 indicates an elimination, bimolecular reaction, where rate = k [B][R-LG]. This implies that the rate determining step involves an interaction between these two species, the base B, and the organic substrate, R-LG
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
In E2 (not E1) reactions, the eliminated H and LG must be anti-periplanar (anti-coplanar) to each other. In In cyclohexane rings, this means that the eliminated H and LG must both be axial and anti-coplanar .
Geometrical analysis confirmed synchronous E2-type transition states for the elimination of the current reaction, instead of nonconcerted processes through E1cb-like and E1-like mechanisms. Importantly, the controversy concerning the reactant complex for anti-E2 …
Elimination Reactions and Cyclohexane Conformation In E2
Mechanism of Elimination Reactions CliffsNotes
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
5 Stereoselectivity in E2 • Stereoselectivity – mechanistically there is a choice of two products but one is favoured – there is a choice • If two protons can be eliminated the reaction will proceed via the
In this practice problem, you will need to determine the major organic product and the mechanism of each reaction. This covers the competition between S N 1, S N 2 nucleophilic substitution and E1/E2 elimination reactions.
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
E1 Reactions Chemistry LibreTexts
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
5 Stereoselectivity in E2 • Stereoselectivity – mechanistically there is a choice of two products but one is favoured – there is a choice • If two protons can be eliminated the reaction will proceed via the
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
11/11/2016 · You’ll also see the step by step mechanism for what appears to be a tricky intermediate transition. This question came from a student’s exam where over 100 students failed to get full credit
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
The E2 Mechanism-Regioselectivity Attack of base Attack of base “Kinetic” product “Thermodynamic” product. The E2 Mechanism-Regioselectivity ~30% ~70% . The E2 Mechanism-Regioselectivity 2-bromopentane 1-pentene 2-pentene. The E2 Mechanism-Stereoselectivity. Regioselectivity-The tendency of a reaction to preferentially produce one constitutional isomer. Stereoselectivity-The …
In E2 (not E1) reactions, the eliminated H and LG must be anti-periplanar (anti-coplanar) to each other. In In cyclohexane rings, this means that the eliminated H and LG must both be axial and anti-coplanar .
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
28/09/2015 · This video shows you an easy way to identify if a reaction will undergo an SN1, SN2, E1, or E2 mechanism. SN1 reactions generate a racemic mixture of stereoisomers due to the planar structure of


Know the detailed mechanisms of SN1, SN2, E1 and E2 2. Know what is a good nucleophile and what is a poor nucleophile 3. Understand the concept of inversion of configuration (SN2) 4. Know the kinetics associated with each reaction 5. Know Zaitzev¶s Rule and how it applies to the elimination reactions. 6. Know what is a good leaving group in a reaction.
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
Unimolecular elimination reactions, also called E1, use a different mechanism that involves a carbocation. Since primary carbocations rarely form, elimination of a primary alcohol will occur via
Mechanism of Elimination Reactions CliffsNotes
E1 And E2. Conference Paper E2 mechanism. Strong bases like ¯OH and ¯OR favor E2 reactions, whereas . weaker bases like H 2 O and ROH favo r E1 reactions. • No kinetic isotopic effect and
1 Stereoselectivity in E1 (E or Z Massey University
Ch 5 E2 mechanism
11/11/2016 · You’ll also see the step by step mechanism for what appears to be a tricky intermediate transition. This question came from a student’s exam where over 100 students failed to get full credit
E1 Reaction Mechanism Organic chemistry
E2 mechanism. E2 indicates an elimination, bimolecular reaction, where rate = k [B][R-LG]. This implies that the rate determining step involves an interaction between these two species, the base B, and the organic substrate, R-LG
Elimination reactions E1 E2 SUNY Geneseo
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
E1 Reactions Chemistry LibreTexts
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
Exam 3 Name CHEM 210 Pennsylvania State University
1 Stereoselectivity in E1 (E or Z Massey University
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
E1 and E2 Reactions Organic Chemistry Socratic
– [Instructor] Let’s look at the mechanism for an E1 elimination reaction, and we’ll start with our substrate, so on the left. Let’s say we’re dealing with alkyl halide. So the carbon that’s bonded to our halogen would be the alpha carbon, and the carbon next to that carbon would be the beta carbon
E2 Elimination & Mechanism Reaction Definition & Examples
ANSWERS 1) Br CH3 CH3 ethanol D CH3 CH3 1) +Br 2) CH3 CH3 rearrangement CH3 CH3 E1 mechanism with carbocation rearrangement 3) CH 3 H3C H HOCH2CH3 CH3 CH+H 2OCH2CH3 2) In questions 2 and 3, only the proton trans to the leaving group can eliminate.
1 Stereoselectivity in E1 (E or Z Massey University
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
E1 AND E2 REACTIONS EPUB DOWNLOAD – (Pdf Plus.) The rate of the E1 reaction depends only on the substrate, since the rate limiting step is the formation of a carbocation. Hence, the more stable that carbocation is, the faster the reaction will be. The first case is an E2 reaction.
1 Stereoselectivity in E1 (E or Z Massey University
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
Elimination Reactions and Cyclohexane Conformation In E2
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
E1 Reaction Mechanism Organic chemistry
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
E2 Reactions
E1 and E2 reactions Chemical Reactions Organic Chemistry
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
Mechanism of Elimination Reactions CliffsNotes
E1 and E2 Reactions Organic Chemistry Socratic
Elimination reactions E1 E2 SUNY Geneseo
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
E1 Reactions Chemistry LibreTexts
Geometrical analysis confirmed synchronous E2-type transition states for the elimination of the current reaction, instead of nonconcerted processes through E1cb-like and E1-like mechanisms. Importantly, the controversy concerning the reactant complex for anti-E2 …
E1 mechanism kinetics and substrate (video) Khan Academy
1 Stereoselectivity in E1 (E or Z Massey University
Elimination Reactions and Cyclohexane Conformation In E2
There are three versions of an elimination reaction: E1, E2 and E1cB! (the E1cB mechanism is very rare and only occurs under very select conditions)!
Elimination reactions E1 E2 SUNY Geneseo
(PDF) E1 And E2 ResearchGate
This organic chemistry video tutorial provides a basic introduction into SN2, SN1, E1 and E2 reaction mechanisms. It provides a chart to determine which reaction mechanism will yield the major product given a particular alkyl halide or substrate.
Elimination Reactions Department of Chemistry
E1 mechanism kinetics and substrate (video) Khan Academy
– [Instructor] Let’s look at the mechanism for an E1 elimination reaction, and we’ll start with our substrate, so on the left. Let’s say we’re dealing with alkyl halide. So the carbon that’s bonded to our halogen would be the alpha carbon, and the carbon next to that carbon would be the beta carbon
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
Elimination Reactions Organic Chemistry – Lecture Notes
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
E2 Reactions
E1 AND E2 REACTIONS EPUB DOWNLOAD – (Pdf Plus.) The rate of the E1 reaction depends only on the substrate, since the rate limiting step is the formation of a carbocation. Hence, the more stable that carbocation is, the faster the reaction will be. The first case is an E2 reaction.
Elimination Reactions Organic Chemistry – Lecture Notes
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
E2 Reactions
The E2 Mechanism-Regioselectivity Attack of base Attack of base “Kinetic” product “Thermodynamic” product. The E2 Mechanism-Regioselectivity ~30% ~70% . The E2 Mechanism-Regioselectivity 2-bromopentane 1-pentene 2-pentene. The E2 Mechanism-Stereoselectivity. Regioselectivity-The tendency of a reaction to preferentially produce one constitutional isomer. Stereoselectivity-The …
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
5.16 Anti Elimination in E2 Reactions Stereoelectronic
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
The E2 Mechanism-Regioselectivity Attack of base Attack of base “Kinetic” product “Thermodynamic” product. The E2 Mechanism-Regioselectivity ~30% ~70% . The E2 Mechanism-Regioselectivity 2-bromopentane 1-pentene 2-pentene. The E2 Mechanism-Stereoselectivity. Regioselectivity-The tendency of a reaction to preferentially produce one constitutional isomer. Stereoselectivity-The …
5.16 Anti Elimination in E2 Reactions Stereoelectronic
11/11/2016 · You’ll also see the step by step mechanism for what appears to be a tricky intermediate transition. This question came from a student’s exam where over 100 students failed to get full credit
Elimination reactions E1 E2 SUNY Geneseo
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
Ch 5 E2 mechanism
E1 and E2 reactions Chemical Reactions Organic Chemistry
Ace Organic Chemistry Reactions & Mechanisms with E.A.S.E. Organic Chemistry Reactions Chemistry Help Chemistry Notes Science Chemistry Physical Science Science Education Science Lessons Science Experiments Teaching Science Knowledge Learning Organic Chemistry Physics Education:__cat__ Education School Science Classroom. Ace Organic Chemistry Reactions & Mechanisms …
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
Elimination Reactions Organic Chemistry – Lecture Notes
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
(PDF) E1 And E2 ResearchGate
Exam 3 Name CHEM 210 Pennsylvania State University
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
Geometrical analysis confirmed synchronous E2-type transition states for the elimination of the current reaction, instead of nonconcerted processes through E1cb-like and E1-like mechanisms. Importantly, the controversy concerning the reactant complex for anti-E2 …
E2 Reactions
E1 and E2 Reactions Organic Chemistry Socratic
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
Mechanism of Elimination Reactions CliffsNotes
Elimination Reactions Department of Chemistry
E1 Reactions Chemistry LibreTexts
Know the detailed mechanisms of SN1, SN2, E1 and E2 2. Know what is a good nucleophile and what is a poor nucleophile 3. Understand the concept of inversion of configuration (SN2) 4. Know the kinetics associated with each reaction 5. Know Zaitzev¶s Rule and how it applies to the elimination reactions. 6. Know what is a good leaving group in a reaction.
5.16 Anti Elimination in E2 Reactions Stereoelectronic
E1 Reactions Chemistry LibreTexts
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
Exam 3 Name CHEM 210 Pennsylvania State University
E2 Reactions
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
(PDF) E1 And E2 ResearchGate
Elimination Reactions Organic Chemistry – Lecture Notes
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
Ch 5 E2 mechanism
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
Elimination reactions: E1, E2 E2: biomolecular elimination Reverse of H-X addition Need an a proton if more than one, product mixture results kinetics:rate = k[RBr][B:-] Zaitsev’s Rule: In general, for base promoted elimination reactions, the more highly substituted alkene is the major product. C C H 3C CH 3 H H H Cl B:-b a + loss of a (minor) loss of b (major) CH 3 H H H CH 3 H + H loss of
1 Stereoselectivity in E1 (E or Z Massey University
E2 Elimination & Mechanism Reaction Definition & Examples
5.16 Anti Elimination in E2 Reactions Stereoelectronic
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
E1 Reaction Mechanism Organic chemistry
Exam 3 Name CHEM 210 Pennsylvania State University
In this practice problem, you will need to determine the major organic product and the mechanism of each reaction. This covers the competition between S N 1, S N 2 nucleophilic substitution and E1/E2 elimination reactions.
E1 mechanism kinetics and substrate (video) Khan Academy
Elimination reactions: E1, E2 E2: biomolecular elimination Reverse of H-X addition Need an a proton if more than one, product mixture results kinetics:rate = k[RBr][B:-] Zaitsev’s Rule: In general, for base promoted elimination reactions, the more highly substituted alkene is the major product. C C H 3C CH 3 H H H Cl B:-b a + loss of a (minor) loss of b (major) CH 3 H H H CH 3 H + H loss of
Elimination reactions E1 E2 SUNY Geneseo
1 Stereoselectivity in E1 (E or Z Massey University
E1 and E2 Reactions Organic Chemistry Socratic
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
E1 Reactions Chemistry LibreTexts
E2 Reactions
– [Instructor] Let’s look at the mechanism for an E1 elimination reaction, and we’ll start with our substrate, so on the left. Let’s say we’re dealing with alkyl halide. So the carbon that’s bonded to our halogen would be the alpha carbon, and the carbon next to that carbon would be the beta carbon
Ch 5 E2 mechanism
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
Ch 5 E2 mechanism
E1 and E2 Reactions Organic Chemistry Socratic
E1 and E2 reactions Chemical Reactions Organic Chemistry
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
E1 and E2 Reactions Organic Chemistry Socratic
Ch 5 E2 mechanism
5.16 Anti Elimination in E2 Reactions Stereoelectronic
Chapter 11: Reactions of Alkyl Halides Coverage: 1. Substitution Reactions, SN1 and SN2 2. Elimination Reactions, E1 and E2 Problems:25-39, 43,47,54 Goals: 1. Know the detailed… Substitution Reactions, SN1 and SN2 2.
Elimination Reactions and Cyclohexane Conformation In E2
E1 Reactions Chemistry LibreTexts
Elimination reactions E1 E2 SUNY Geneseo
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
E1 and E2 Reactions Organic Chemistry Socratic
Elimination reactions E1 E2 SUNY Geneseo
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
Elimination Reactions Organic Chemistry – Lecture Notes
– [Instructor] Let’s look at the mechanism for an E1 elimination reaction, and we’ll start with our substrate, so on the left. Let’s say we’re dealing with alkyl halide. So the carbon that’s bonded to our halogen would be the alpha carbon, and the carbon next to that carbon would be the beta carbon
Ch 5 E2 mechanism
E2 Reactions
E1 and E2 reactions Chemical Reactions Organic Chemistry
E1 And E2. Conference Paper E2 mechanism. Strong bases like ¯OH and ¯OR favor E2 reactions, whereas . weaker bases like H 2 O and ROH favo r E1 reactions. • No kinetic isotopic effect and
E2 Elimination & Mechanism Reaction Definition & Examples
Elimination Reactions Organic Chemistry – Lecture Notes
There are three versions of an elimination reaction: E1, E2 and E1cB! (the E1cB mechanism is very rare and only occurs under very select conditions)!
1 Stereoselectivity in E1 (E or Z Massey University
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
Exam 3 Name CHEM 210 Pennsylvania State University
E2 Mechanism With primary alcohols we have the E2 mechanism. The “2” indicates that the reaction is bi-molecular in the rate determining step. There are two steps overall. Step 1: The first step is the same as in the first step of the E1 reaction. It is the reversible protonation of the alcohol oxygen to make it into a good leaving group.
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
Elimination Reactions Organic Chemistry – Lecture Notes
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
Elimination Reactions and Cyclohexane Conformation In E2
E1 mechanism kinetics and substrate (video) Khan Academy
E2 Reactions
11/11/2016 · You’ll also see the step by step mechanism for what appears to be a tricky intermediate transition. This question came from a student’s exam where over 100 students failed to get full credit
Elimination reactions E1 E2 SUNY Geneseo
Ch 5 E2 mechanism
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
Unimolecular elimination reactions, also called E1, use a different mechanism that involves a carbocation. Since primary carbocations rarely form, elimination of a primary alcohol will occur via
E1 and E2 Reactions Organic Chemistry Socratic
Exam 3 Name CHEM 210 Pennsylvania State University
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
Mechanism of Elimination Reactions CliffsNotes
E1 Reactions Chemistry LibreTexts
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
– [Instructor] Let’s look at the mechanism for an E1 elimination reaction, and we’ll start with our substrate, so on the left. Let’s say we’re dealing with alkyl halide. So the carbon that’s bonded to our halogen would be the alpha carbon, and the carbon next to that carbon would be the beta carbon
Elimination Reactions and Cyclohexane Conformation In E2
Mechanism of Elimination Reactions CliffsNotes
E1 and E2 Reactions Organic Chemistry Socratic
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
Elimination Reactions and Cyclohexane Conformation In E2
Elimination Reactions Department of Chemistry
Elimination Reactions Organic Chemistry – Lecture Notes
Geometrical analysis confirmed synchronous E2-type transition states for the elimination of the current reaction, instead of nonconcerted processes through E1cb-like and E1-like mechanisms. Importantly, the controversy concerning the reactant complex for anti-E2 …
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
Know the detailed mechanisms of SN1, SN2, E1 and E2 2. Know what is a good nucleophile and what is a poor nucleophile 3. Understand the concept of inversion of configuration (SN2) 4. Know the kinetics associated with each reaction 5. Know Zaitzev¶s Rule and how it applies to the elimination reactions. 6. Know what is a good leaving group in a reaction.
1 Stereoselectivity in E1 (E or Z Massey University
ANSWERS 1) Br CH3 CH3 ethanol D CH3 CH3 1) +Br 2) CH3 CH3 rearrangement CH3 CH3 E1 mechanism with carbocation rearrangement 3) CH 3 H3C H HOCH2CH3 CH3 CH+H 2OCH2CH3 2) In questions 2 and 3, only the proton trans to the leaving group can eliminate.
Elimination Reactions Organic Chemistry – Lecture Notes
E2 Elimination & Mechanism Reaction Definition & Examples
E1 Reaction Mechanism Organic chemistry
The mechanism of the following reaction is a) SN1 b) SN2 c) E1 d) E2 Predict the product(s) for the following reaction. Consider the reactions shown and answer the associated questions.
Mechanism of Elimination Reactions CliffsNotes
E1 Reactions Chemistry LibreTexts
Ch 5 E2 mechanism
E2 eliminations, in contrast to E1 reactions are promoted by strong base. The base vital to the reaction; it is directly involved in the rate-determining step. The reaction is bimolecular–that is, it involves “second-order kinetics–because two molecules must come together for the reaction to occur. The mechanism of an E2 elimination reaction is shown below:
Exam 3 Name CHEM 210 Pennsylvania State University
5.16 Anti Elimination in E2 Reactions Stereoelectronic
The E2 Mechanism-Regioselectivity Attack of base Attack of base “Kinetic” product “Thermodynamic” product. The E2 Mechanism-Regioselectivity ~30% ~70% . The E2 Mechanism-Regioselectivity 2-bromopentane 1-pentene 2-pentene. The E2 Mechanism-Stereoselectivity. Regioselectivity-The tendency of a reaction to preferentially produce one constitutional isomer. Stereoselectivity-The …
Elimination Reactions and Cyclohexane Conformation In E2
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
E2 Elimination & Mechanism Reaction Definition & Examples
Elimination Reactions Department of Chemistry
E1 Reaction Mechanism Organic chemistry
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
Mechanism of Elimination Reactions CliffsNotes
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
Ch 5 E2 mechanism
Mechanism of Elimination Reactions CliffsNotes
Elimination Reactions and Cyclohexane Conformation In E2
Geometrical analysis confirmed synchronous E2-type transition states for the elimination of the current reaction, instead of nonconcerted processes through E1cb-like and E1-like mechanisms. Importantly, the controversy concerning the reactant complex for anti-E2 …
E1 and E2 Reactions Organic Chemistry Socratic
E1 mechanism kinetics and substrate (video) Khan Academy
E1 and E2 reactions Chemical Reactions Organic Chemistry
This organic chemistry video tutorial provides a basic introduction into SN2, SN1, E1 and E2 reaction mechanisms. It provides a chart to determine which reaction mechanism will yield the major product given a particular alkyl halide or substrate.
Exam 3 Name CHEM 210 Pennsylvania State University
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
Ace Organic Chemistry Reactions & Mechanisms with E.A.S.E. Organic Chemistry Reactions Chemistry Help Chemistry Notes Science Chemistry Physical Science Science Education Science Lessons Science Experiments Teaching Science Knowledge Learning Organic Chemistry Physics Education:__cat__ Education School Science Classroom. Ace Organic Chemistry Reactions & Mechanisms …
Ch 5 E2 mechanism
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
E1 Reactions Chemistry LibreTexts
In E2 (not E1) reactions, the eliminated H and LG must be anti-periplanar (anti-coplanar) to each other. In In cyclohexane rings, this means that the eliminated H and LG must both be axial and anti-coplanar .
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
E1 mechanism kinetics and substrate (video) Khan Academy
E1 and E2 reactions Chemical Reactions Organic Chemistry
Unimolecular elimination reactions, also called E1, use a different mechanism that involves a carbocation. Since primary carbocations rarely form, elimination of a primary alcohol will occur via
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
E2 Reactions
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
Mechanism of Elimination Reactions CliffsNotes
Exam 3 Name CHEM 210 Pennsylvania State University
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
Elimination Reactions Department of Chemistry
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
Elimination Reactions Department of Chemistry
E1 Reaction Mechanism Organic chemistry
Unlike E2 reactions, which require the proton to be anti to the leaving group, E1 reactions only require a neighboring hydrogen. This is due to the fact that the leaving group has already left the molecule. The final product is an alkene along with the HB byproduct.
E1 mechanism kinetics and substrate (video) Khan Academy
E1 Reactions Chemistry LibreTexts
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
Elimination Reactions Organic Chemistry – Lecture Notes
E1 Reactions Chemistry LibreTexts
Ch 5 E2 mechanism
5 Stereoselectivity in E2 • Stereoselectivity – mechanistically there is a choice of two products but one is favoured – there is a choice • If two protons can be eliminated the reaction will proceed via the
Elimination Reactions Organic Chemistry – Lecture Notes
E1 mechanism kinetics and substrate (video) Khan Academy
ANSWERS 1) Br CH3 CH3 ethanol D CH3 CH3 1) +Br 2) CH3 CH3 rearrangement CH3 CH3 E1 mechanism with carbocation rearrangement 3) CH 3 H3C H HOCH2CH3 CH3 CH+H 2OCH2CH3 2) In questions 2 and 3, only the proton trans to the leaving group can eliminate.
E1 Reaction Mechanism Organic chemistry
E1 AND E2 REACTIONS EPUB DOWNLOAD – (Pdf Plus.) The rate of the E1 reaction depends only on the substrate, since the rate limiting step is the formation of a carbocation. Hence, the more stable that carbocation is, the faster the reaction will be. The first case is an E2 reaction.
E1 and E2 Reactions Organic Chemistry Socratic
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
E2 Reactions
E1 and E2 reactions Chemical Reactions Organic Chemistry
E1 Reaction Mechanism Organic chemistry
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
Elimination Reactions and Cyclohexane Conformation In E2
The E2 Mechanism-Regioselectivity Attack of base Attack of base “Kinetic” product “Thermodynamic” product. The E2 Mechanism-Regioselectivity ~30% ~70% . The E2 Mechanism-Regioselectivity 2-bromopentane 1-pentene 2-pentene. The E2 Mechanism-Stereoselectivity. Regioselectivity-The tendency of a reaction to preferentially produce one constitutional isomer. Stereoselectivity-The …
Elimination Reactions Department of Chemistry
E1 and E2 Reactions Organic Chemistry Socratic
E1 and E2 reactions Chemical Reactions Organic Chemistry
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
Elimination Reactions Organic Chemistry – Lecture Notes
Mechanism of Elimination Reactions CliffsNotes
E1 mechanism kinetics and substrate (video) Khan Academy
28/09/2015 · This video shows you an easy way to identify if a reaction will undergo an SN1, SN2, E1, or E2 mechanism. SN1 reactions generate a racemic mixture of stereoisomers due to the planar structure of
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
The mechanism of the following reaction is a) SN1 b) SN2 c) E1 d) E2 Predict the product(s) for the following reaction. Consider the reactions shown and answer the associated questions.
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
E1 mechanism kinetics and substrate (video) Khan Academy
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
Elimination reactions E1 E2 SUNY Geneseo
E1 reactions are faster if the intermediate carbocation is more stable. By the Hammond postulate, the transition state lowers in By the Hammond postulate, the transition state lowers in energy in an endothermic process if the products are more stable.
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
1 Stereoselectivity in E1 (E or Z Massey University
E1 Reaction Mechanism Organic chemistry
Overview: The general form of the E1 mechanism is as follows: B: = base X = leaving group (usually halide or tosylate) In the E1 mechanism, the the first step is the loss of the leaving group, which leaves in a very slow step, resulting in the formation of a carbocation.
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
Elimination reactions E1 E2 SUNY Geneseo
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
1 Stereoselectivity in E1 (E or Z Massey University
Ch 5 E2 mechanism
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
Elimination Reactions Organic Chemistry – Lecture Notes
E2 mechanism. E2 indicates an elimination, bimolecular reaction, where rate = k [B][R-LG]. This implies that the rate determining step involves an interaction between these two species, the base B, and the organic substrate, R-LG
E2 Elimination & Mechanism Reaction Definition & Examples
In this practice problem, you will need to determine the major organic product and the mechanism of each reaction. This covers the competition between S N 1, S N 2 nucleophilic substitution and E1/E2 elimination reactions.
Ch 5 E2 mechanism
Elimination Reactions Organic Chemistry – Lecture Notes
Elimination Reactions and Cyclohexane Conformation In E2
E2 Mechanism With primary alcohols we have the E2 mechanism. The “2” indicates that the reaction is bi-molecular in the rate determining step. There are two steps overall. Step 1: The first step is the same as in the first step of the E1 reaction. It is the reversible protonation of the alcohol oxygen to make it into a good leaving group.
Exam 3 Name CHEM 210 Pennsylvania State University
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
Ch 5 E2 mechanism
Elimination Reactions Department of Chemistry
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
(PDF) E1 And E2 ResearchGate
E1 Reaction Mechanism Organic chemistry
1 Stereoselectivity in E1 (E or Z Massey University
11/11/2016 · You’ll also see the step by step mechanism for what appears to be a tricky intermediate transition. This question came from a student’s exam where over 100 students failed to get full credit
Mechanism of Elimination Reactions CliffsNotes
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
Unimolecular elimination reactions, also called E1, use a different mechanism that involves a carbocation. Since primary carbocations rarely form, elimination of a primary alcohol will occur via
1 Stereoselectivity in E1 (E or Z Massey University
E1 mechanism kinetics and substrate (video) Khan Academy
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
Elimination reactions E1 E2 SUNY Geneseo
(PDF) E1 And E2 ResearchGate
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
(PDF) E1 And E2 ResearchGate
1, E2, E1) yielded different products Trends to predict which mechanism will predominate 1) Weakly basic species that are good nucleophiles give predominantly substitution
1 Stereoselectivity in E1 (E or Z Massey University
Elimination Reactions and Cyclohexane Conformation In E2
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
E1 Reactions Chemistry LibreTexts
Ch 5 E2 mechanism
Elimination Reactions and Cyclohexane Conformation In E2
28/09/2015 · This video shows you an easy way to identify if a reaction will undergo an SN1, SN2, E1, or E2 mechanism. SN1 reactions generate a racemic mixture of stereoisomers due to the planar structure of
E1 Reaction Mechanism Organic chemistry
E2 Reactions
Elimination reactions E1 E2 SUNY Geneseo
Chapter 10 November 15, 2000 Page – 1 Elimination Reactions, E2 The E2 elimination is a concerted reaction involving the deprotonation of a carbon adjacent to a carbon bearing a good leaving group.
E2 Reactions
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
E1 mechanism kinetics and substrate (video) Khan Academy
There are three versions of an elimination reaction: E1, E2 and E1cB! (the E1cB mechanism is very rare and only occurs under very select conditions)!
Exam 3 Name CHEM 210 Pennsylvania State University
Elimination reactions E1 E2 SUNY Geneseo
Elimination reactions: E1, E2 E2: biomolecular elimination Reverse of H-X addition Need an a proton if more than one, product mixture results kinetics:rate = k[RBr][B:-] Zaitsev’s Rule: In general, for base promoted elimination reactions, the more highly substituted alkene is the major product. C C H 3C CH 3 H H H Cl B:-b a + loss of a (minor) loss of b (major) CH 3 H H H CH 3 H + H loss of
Mechanism of Elimination Reactions CliffsNotes
E2 Elimination & Mechanism Reaction Definition & Examples
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
Elimination Reactions Organic Chemistry – Lecture Notes
5.16 Anti Elimination in E2 Reactions Stereoelectronic
E1 Reaction Mechanism Organic chemistry
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
Elimination Reactions Organic Chemistry – Lecture Notes
– [Instructor] Let’s look at the mechanism for an E1 elimination reaction, and we’ll start with our substrate, so on the left. Let’s say we’re dealing with alkyl halide. So the carbon that’s bonded to our halogen would be the alpha carbon, and the carbon next to that carbon would be the beta carbon
1 Stereoselectivity in E1 (E or Z Massey University
E1 and E2 Reactions Organic Chemistry Socratic
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
E1 Reactions Chemistry LibreTexts
In this practice problem, you will need to determine the major organic product and the mechanism of each reaction. This covers the competition between S N 1, S N 2 nucleophilic substitution and E1/E2 elimination reactions.
E1 and E2 Reactions Organic Chemistry Socratic
Watch Next Video: E1 Reaction Rate and Mechanism – Unimolecular beta-elimination Part 1 Watch Previous Video: Choosing Between SN1 and SN2 Reactions Part 1 This is video 15 in the SN1 SN2 E1 E2 …
(PDF) E1 And E2 ResearchGate
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
Elimination Reactions Department of Chemistry
At even modest concentrations of strong base, elimination by the E2 mechanism is much faster than E1 elimination. There is a strong similarity between the mechanism shown in Figure 5.12 and the one shown for alcohol dehydration in Figure 5.6.
E1 and E2 reactions [PPT Powerpoint] – vdocuments.mx
Elimination Reactions and Cyclohexane Conformation In E2
This organic chemistry video tutorial provides a basic introduction into SN2, SN1, E1 and E2 reaction mechanisms. It provides a chart to determine which reaction mechanism will yield the major product given a particular alkyl halide or substrate.
E1 and E2 reactions Chemical Reactions Organic Chemistry
1 Stereoselectivity in E1 (E or Z Massey University
In this practice problem, you will need to determine the major organic product and the mechanism of each reaction. This covers the competition between S N 1, S N 2 nucleophilic substitution and E1/E2 elimination reactions.
Elimination reactions E1 E2 SUNY Geneseo
Ace Organic Chemistry Reactions & Mechanisms with E.A.S.E. Organic Chemistry Reactions Chemistry Help Chemistry Notes Science Chemistry Physical Science Science Education Science Lessons Science Experiments Teaching Science Knowledge Learning Organic Chemistry Physics Education:__cat__ Education School Science Classroom. Ace Organic Chemistry Reactions & Mechanisms …
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
E1 mechanism kinetics and substrate (video) Khan Academy
Elimination reactions (E1 and E2) where E2 is one step and E1 is two steps. Both result in the removal of two substituents and E1 is a carbocation reaction. Both result in the removal of two substituents and E1 is a carbocation reaction.
E1 Reactions Chemistry LibreTexts
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
Elimination Reactions Department of Chemistry
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
Chapter 11: Reactions of Alkyl Halides Coverage: 1. Substitution Reactions, SN1 and SN2 2. Elimination Reactions, E1 and E2 Problems:25-39, 43,47,54 Goals: 1. Know the detailed… Substitution Reactions, SN1 and SN2 2.
E1 Reactions Chemistry LibreTexts
Elimination Reactions Organic Chemistry – Lecture Notes
Exam 3 Name CHEM 210 Pennsylvania State University
Weak base favors E1 reaction by disfavoring E2 reaction Not effected but a low concentration of base favors E1 by disfavoring a E2 reaction Protic polar favors a E1 reaction if the reactant is not charged. Bulkiest groups on opposite sides SN Versus E Methyl halide Primary halide Secondary halide Tertiary halide SN2 reaction most favored No Elimination reactions! SN2 when the main reaction is
Elimination Reactions and Cyclohexane Conformation In E2
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
Ch 5 E2 mechanism
Unimolecular elimination reactions, also called E1, use a different mechanism that involves a carbocation. Since primary carbocations rarely form, elimination of a primary alcohol will occur via
E1 Reactions Chemistry LibreTexts
Elimination Reactions Organic Chemistry – Lecture Notes
The actual mechanism can be one of two types, E1 or E2, depending upon the structure of the activated complex. E1 mechanism An atom that bears a …
1 Stereoselectivity in E1 (E or Z Massey University
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
Mechanism of Elimination Reactions CliffsNotes
ANSWERS 1) Br CH3 CH3 ethanol D CH3 CH3 1) +Br 2) CH3 CH3 rearrangement CH3 CH3 E1 mechanism with carbocation rearrangement 3) CH 3 H3C H HOCH2CH3 CH3 CH+H 2OCH2CH3 2) In questions 2 and 3, only the proton trans to the leaving group can eliminate.
Elimination Reactions Organic Chemistry – Lecture Notes
E1 mechanism kinetics and substrate (video) Khan Academy
5.16 Anti Elimination in E2 Reactions Stereoelectronic
ELIMINATION REACTIONS. Objective and Outline beta-eliminations E1, E2 . and . E1. c. B. mechanisms Stereochemical considerations of these reactions
Mechanism of Elimination Reactions CliffsNotes
Think about the mechanism of each reaction. What is the mechanism of each reaction, and are the intermediates, or the transition states involved? What effect do you think the polarity and the protonic/non-protonic nature of the solvent would have
Ch 5 E2 mechanism
5 Stereoselectivity in E2 • Stereoselectivity – mechanistically there is a choice of two products but one is favoured – there is a choice • If two protons can be eliminated the reaction will proceed via the
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
E2 Reactions
E1 Reaction Mechanism Organic chemistry
Weak base favors E1 reaction by disfavoring E2 reaction Not effected but a low concentration of base favors E1 by disfavoring a E2 reaction Protic polar favors a E1 reaction if the reactant is not charged. Bulkiest groups on opposite sides SN Versus E Methyl halide Primary halide Secondary halide Tertiary halide SN2 reaction most favored No Elimination reactions! SN2 when the main reaction is
SparkNotes Organic Chemistry Sn1E1 Reactions SN1 and E1
Chapter 11: Reactions of Alkyl Halides Coverage: 1. Substitution Reactions, SN1 and SN2 2. Elimination Reactions, E1 and E2 Problems:25-39, 43,47,54 Goals: 1. Know the detailed… Substitution Reactions, SN1 and SN2 2.
1 Stereoselectivity in E1 (E or Z Massey University
Unimolecular elimination reactions, also called E1, use a different mechanism that involves a carbocation. Since primary carbocations rarely form, elimination of a primary alcohol will occur via
E1 Reactions Chemistry LibreTexts
Elimination Reactions Organic Chemistry – Lecture Notes
E2 Elimination & Mechanism Reaction Definition & Examples
A summary of SN1 and E1 Reactions in ‘s Organic Chemistry: Sn1E1 Reactions. Learn exactly what happened in this chapter, scene, or section of Organic Chemistry: Sn1E1 Reactions and what it means. Perfect for acing essays, tests, and quizzes, as well as for writing lesson plans.
E2 Reactions
Elimination reactions E1 E2 SUNY Geneseo
E1 mechanism kinetics and substrate (video) Khan Academy
Elimination Reactions and Cyclohexane Conformation In E2 reactions in cyclohexane rings, the anti-periplanar orientation is possible only if the halogen and the H are 1,2 trans-diaxial. For cyclohexanes that are conformationally biased (one chair form strongly favored over the other), the differences between the rates of reaction of the two isomers can be large.
1 Stereoselectivity in E1 (E or Z Massey University
Know the detailed mechanisms of SN1, SN2, E1 and E2 2. Know what is a good nucleophile and what is a poor nucleophile 3. Understand the concept of inversion of configuration (SN2) 4. Know the kinetics associated with each reaction 5. Know Zaitzev¶s Rule and how it applies to the elimination reactions. 6. Know what is a good leaving group in a reaction.
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
E2 Mechanism With primary alcohols we have the E2 mechanism. The “2” indicates that the reaction is bi-molecular in the rate determining step. There are two steps overall. Step 1: The first step is the same as in the first step of the E1 reaction. It is the reversible protonation of the alcohol oxygen to make it into a good leaving group.
E2 Reactions
Elimination Reactions Department of Chemistry
E2 Mechanism With primary alcohols we have the E2 mechanism. The “2” indicates that the reaction is bi-molecular in the rate determining step. There are two steps overall. Step 1: The first step is the same as in the first step of the E1 reaction. It is the reversible protonation of the alcohol oxygen to make it into a good leaving group.
Elimination Reactions and Cyclohexane Conformation In E2
Elimination Reactions Organic Chemistry – Lecture Notes
E2 Elimination & Mechanism Reaction Definition & Examples
Chapter 11: Reactions of Alkyl Halides Coverage: 1. Substitution Reactions, SN1 and SN2 2. Elimination Reactions, E1 and E2 Problems:25-39, 43,47,54 Goals: 1. Know the detailed… Substitution Reactions, SN1 and SN2 2.
E1 AND E2 REACTIONS EPUB DOWNLOAD (Pdf Plus.)
SN2 SN1 E1 E2 Reaction Mechanisms Made Easy! VidioHD
staggered conformation is more favorable for E2 reactions (unlike E1 reactions). Elimination reaction of cyclohexanol to cyclohexene with sulfuric acid and heat [1] An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one or two-step mechanism.[2] The one-step mechanism is known as the E2 reaction, and the two-step mechanism …
E1 and E2 reactions Chemical Reactions Organic Chemistry
1 Stereoselectivity in E1 (E or Z Massey University
E1 mechanism kinetics and substrate (video) Khan Academy