Solvent effects on reaction rates and mechanisms pdf
Solvents effects on reaction rates Solvents can affect rates through equilibrium-solvent effects that can be explained on the basis of the transition state theory. In essence, the reaction rates are influenced by differential solvation of the starting material and transition state by the solvent.
The rates of formal abstraction of phenolic hydrogen atoms by free radicals, Y • + ArOH → YH + ArO •, are profoundly influenced by the hydrogen-bond-accepting and anion-solvation abilities of solvents, by the electron affinities and reactivities (Y−H bond dissociation enthalpies) of radicals, and by the phenol’s ring substituents.
Eur. J. Biochem. 176, 273-279 (1988) 0 FEBS 1988 Effect of ‘solvent, pressure and temperature on reaction rates of the multiheme hydroxylamine oxidoreductase
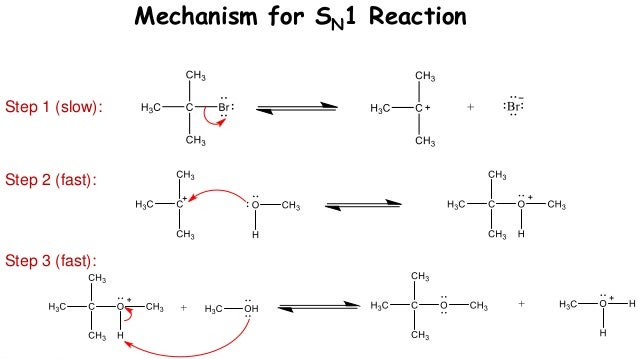
Below is the same reaction conducted in two different solvents and the relative rate that corresponds with it. The figure below shows the mechanism of an S N 1 reaction of an alkyl halide with water. Since water is also the solvent, this is an example of a solvolysis reaction.
Solvent Effects. The nature of the solvent can also affect the reaction rates of solute particles. For example, a sodium acetate solution reacts with methyl iodide in an exchange reaction to give methyl acetate and sodium iodide.
In this study, the mechanism of eleven thiol-ene reaction has been studied by using Density In contrast to the general opinion, radicals are responsive to polar solvents [36].
Unlike S N 1, S N 2 represents bimolecular reactions, and the rate of reaction can be expressed by, rate = K’ [R-LG] [Nu –]. In addition, S N 1 pathway is a multi-step process, and S N 2 pathway is a single step process.
step reaction mechanism (vide infra) and the rate expression is given by Eqn 2 where kf is the rate constant for the fluctuation-limited solvent rearrangement [8]. Such a …
Solvent effects on the reactions of the extremely toxic nerve agent VX with KF/Al2O3 powder were explored. Small quantities of water or methanol (5-10 wt %), which effectively mobilized all components while maintaining the heterogeneous nature of the reaction, promoted much faster rates than those obtained with larger quantities.
SOLVENT EFFECTS IN CIS-TRANS ISOMERIZATION

Facets of Coordination Chemistry SOLVENT EFFECTS ON THE
theory, the solvent effect (ratio of rates in gas phase to those in aqueous phase) can be reduced to an expression involving only the solvation free energies of reactants and transition state. Then,
SN2 , SN1 , E2 , & E1: Substitution and Elimination Reactions l Nucleophilic Substitution Reactions – SN2 Reaction: • Reaction is: o Stereospecific (Walden Inversion of configuration) o Concerted – all bonds form and break at same time o Bimolecular – rate depends on concentration of both nucleophile and substrate • Substrate: o Best if primary (one substituent on carbon bearing leaving
conclusion as to how the nature of the solvent effects the reaction rate and how this relates to the mechanism based on your results. After completing the above, compare your results with the other gamma groups, reconcile any
Noncovalent interactions play a central role in chemical structure and reactivity. Chemical reaction rates are influenced strongly by nonbonding interactions, particularly the solvation processes that evolve as reactants approach, and the short-range repulsions that constrain motion through the transition state.
and MPWB1K) are used to study solvent effects on two types of reaction mechanisms: concerted non-ionic and stepwise ionic. The ionic mechanism is the one found in most organic chemistry
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for the reactions involving the light (k L) and the heavy (k H) isotopically substituted reactants
The solvent effect on reaction rate are best treated in terms of the thermodynamics of hypothetical equilibrium between reactant and transition state, i.e., in the frame of the absolute

Angewandte Chemie DOI: 10.1002/anie.201408359 Biomass Conversion Solvent Effects in Acid-Catalyzed Biomass Conversion Reactions** Max A. Mellmer, Canan Sener, Jean Marcel R. Gallo, Jeremy S. Luterbacher, David Martin Alonso, and James A. Dumesic* Abstract: Reaction kinetics were studied to quantify the effects biomass.[5] Other polar aprotic solvents, such as g-lactones and of …
THE EFFECT OF SOLVENTS UPON EQUILIBRIA, SPECTRA, AND REACTION RATES’ ABSTRACT It is shown that the effect of solvent upon a wide variety of equilibria, reactipn rates, and spectral shifts may be described by the equation log (kao~ventlkothnno~) = SR. S IS a constant related to the polarity of the solvent and R is a measure of the sensitivity of the system to change in solvent polarity. …
reaction mechanism). The correct interpretation of rate data depends on a knowledge of those factors that can influence the rate and these include 1. the structure of the compound or compounds entering into the reaction, 2. the temperature, 3. the type of solvent in which the reaction is carried out, and 4. for many reactions, the concentrations of the reacting species. In this experiment, a
Abstract. Water and mixtures of water with organic solvents are commonly used as solvents for a large variety of organic reactions. Over the last two decades, the mechanisms of many of these processes have been investigated in some detail, usually by kinetic techniques.
A review of polymer dissolution Beth A. Miller-Chou, Jack L. Koenig* Department of Macromolecular Science, Case School Engineering, Case Western Reserve University, 10900 Euclid Avenue, Cleveland, OH 44106, USA Received 13 November 2002 Abstract Polymer dissolution in solvents is an important area of interest in polymer science and engineering because of its many applications in industry such
1/12/2010 · It is common knowledge that solvents have different effects on the rate of degradation, and affect the formation of photoproducts [7, 8]. Moeini-Nombel and Matsuzawa investigated the effect of solvents and substituent groups on the photo-oxidation of fluorene [ 7 ].
Kinetics is the study of how changes in experimental conditions affect the rate of a chosen reaction. Reaction Reaction kinetics can be used to probe the validity of a proposed mechanism.
Solvent Effects on the Reactions of Coordination Complexes 57 solvent induced changes in the potential energy barrier and overall free energy of the reaction.
J. Zhu and J. C. Rasaiah: Electron transfer reactions 9967 to the integral equation. We also discuss how the effect of solvent dynamics on the diffusive part of the ET rate con-

The solvent effect on reaction rate are best treated in terms of the thermodynamics of hypothetical equilibrium between reactant and transition state, i.e., in the frame of the absolute rate theory rather than collision theory [7,8].
For an S N 2 reaction, the effect of solvent polarity is usually much less, but the ability (or really lack there of) of the solvent to solvate the nucleophile is the important criteria, as shown by the relative rate data for the S N 2 reaction of nBuBr with N 3-
Furthermore, for both inner- and outersphere reactions, we show that there is a small solvent effect on the rate of CO 2 insertion. Solvents that have higher acceptor numbers generally lead …
Chem 463 Organic 3 The isotope effect is not limited to the compound under study. Isotopic substitution in the solvent used in the reaction will also give rise to a solvent effect, if the solvent is involved in the reaction.
Catalog Solvent effects on reaction Amis, Edward S. 1905-, Solvent Effects On Reaction Rates and Mechanisms. New York: Academic Press, 1966. MLA Citation (style guide) Amis, Edward S. 1905-. Solvent Effects On Reaction Rates and Mechanisms. New York: Academic Press, 1966. Print. Note! Citation formats are based on standards as of July 2010. Citations contain only title, author, edition
Get this from a library! Solvents effects on reaction rates and mechanisms.. [Edward Stephen Amis]
solvent to the key steps in the reaction mechanisms. One approach to estimating the solvent effect is to addexplicitwatermolecules(∼4perunitcell) 11,18 oreven
9780120573509 Solvent Effects on Reaction Rates and
Polar aprotic solvents increase the rate of E2 reactions. D. Leaving Group Effects The bond to the leaving group is partially broken in the transition state, so the
2 ! !solvent has less of an effect! *Need to compare structures for starting material and transition state for rate determining step, if the amount of charge changes the effect of solvent on reaction rate …
Solvent effects on rates of photochemical reactions of rose bengal triplet state studied by nanosecond laser photolysis Shafiqul D.-M. Islam, Osamu Ito*
Salt effect and common-ion effect: An increase in ionic strength of the solution usually increases the rate of an S N 1reaction.A common ion will deppress the S N 1rate.
Get this from a library! Solvent effects on reaction rates and mechanisms,. [Edward S Amis]
Solvent relaxation effects on the kinetics of photoinduced electron transfer reactions J. Najbar,a) FL C. Dorfman, and M. D. Fayer
To examine further the effect of solvent in such reactions, studies have now been made of the cis-truns isornerizations of azoxyberlzerle and of p,pl-azoxy- toluene in several solvents.
EFFECTS OF SWERCRITICAL SOLVENTS ON THE RATES @F HOMOGENEOUS CHEMICAL REACTIONS K. P Johnston and S. Kim Department of Chemical Engineering – mechanical project engineer resume pdf 1/07/2010 · Introduction. The Menshutkin reaction is regarded as an important example for studying solvent effects upon the rates of reactions; there have been numerous prior experimental 1 and theoretical investigations. 2 Of relevance to this work is kinetic data reported for the Menshutkin reaction between triethylamine and ethyl iodide
392 Bull. Korean Chem. Soc. 1999, Vol. 20, No. 4 Communications to the Editor The Effect of Solvent on Reaction Rates and Equilibria for the Reactions of
1 C X + Nu C X + X X + + or Nu C Nu C Nu Reactions of Alkyl Halides in Which the Bond Between Carbon and Halogen is Broken — An Overview Alkyl halides are prone to undergoing nucleophilic
6/08/2013 · This video gives you a detailed breakdown of the different types of organic solvents you will come across when studying substitution and elimination reactions. By watching this video you will
What Are Kinetic Isotope Effects?! A kinetic isotope effect is a mechanistic phenomenon wherein isotopically substituted molecules react at different rates.
the effects of GVL and other aprotic solvents on the reaction rates for various acid catalysts and reaction temperatures. Table 1 shows the turnover frequencies for reaction 1 (TOF
in considering the solvent effect on the overall reaction rate. The electrophilicity (acidity) of a medium was found to exert the greatest effect on the reaction rate. It is believed that an increase in the electrophilicity is favorable for the rapid protonation of the UC–amine intermediate complex. The effects of amine basicity, ion-ization potential, and dipole moment and the steric
AbeBooks.com: Solvent Effects on Reaction Rates and Mechanisms (9780120573509) by E. S. Amis and a great selection of similar New, Used and Collectible Books available now at great prices.
FACTORS AFFECTING THE RATE OF S N2 REACTIONS 1. Strength of the nucleophile – there are three important trends: 1) For similar species, the species with negative charge is always a stronger nucleophile than a neutral species.
Summary of Solvent Effects on Nucleophilic Substitution Reactions SN1 • Polar solvent stabilizes transition state and carbocation intermediate. • Polar protic solvent makes nucleophile less nucleophilic and stabilizes anionic leaving group. SN2 • Need polar solvent to dissolve nucleophile. • Protic solvent slows rate by solvating nucleophile • Aprotic solvent increases rate by
Effect of solvents on the kinetics and mechanism of the acidic and alkaline hydrolysis of hydroxamic acids Kallol K Ghosh·, The interpretation of kinetic solvent effect on reaction rates is difficult and in general is dominated more by exception than rules. How a particular solvent will effect each step is hard to explain and what is experimentally observed is the net effect. No simple
Solvent effects on the rates and mechanisms of organotin alkyl exchange reactions
Solvent effects on reaction rates 1057 It has therefore been possible, for two particular reactions, to obtain certain transition— state properties by the two main methods involving solvent effects.
Chem 463 Organic University of Victoria
The SN2 Reaction: Mechanism Steric effects in the SN2 reaction: changing the nucleophile concentration does not affect the rate of the reaction! Must be a two-step reaction The overall rate of a reaction is dependent upon the slowest step: rate-limiting step The Mechanism of the SN1 Reaction. 10 step 1 DG‡ step 2 DG‡ˇ step 1 >> DG ‡ step 2 DG‡ˇ step 1 is rate-limiting Reactivity
Reichardt, C. (2002) Solvent Effects on the Rates of Homogeneous Chemical Reactions, in Solvents and Solvent Effects in Organic Chemistry, Third Edition, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, FRG. doi: 10.1002/3527601791.ch5
The Effect of Solvent on Reaction Rates and Mechanisms; 8.Secondary Systems: The Prediction of Reaction Mechanism this occurs by a nucleophilic substitution mechanism , i.e., in which the organic compound reacts with a nucleophile. To do this, the organic molecule must have a good leaving group, which can depart with and stabilize the electron pair of its former bond to carbon. Good
C. the reaction rate increased in the presence of a polar protic solvent D. the reaction showed a rearranged product E. the reaction showed more than one product
effect on the rate and importantly the selectivity of a reaction are at present, difficult to predict for varied reagent/catalyst systems. The effect of the solvent on the rate and selectivity of molecules
The effect of solvent on the rate and mechanism of the reaction was investigated. An inner-sphere mechanism for the reaction was proposed and supported by the calculated activation parameters. An inner-sphere mechanism for the reaction was proposed and …
The reaction intermediate of the SN1 reaction is the carbo-cation whereas the SN2 reaction proceeds through the Transition state. A polar solvent would favour and increase the probability of formation of a charged species i.e the cation.
This study focuses on the Baeyer–Villiger reaction of propanone and performic acid, with formic acid as catalyst. Continuum solvation methods (EIF-PCM and CPCM) and two density functionals (B3LYP and MPWB1K) are used to study solvent effects on two types of reaction mechanisms: concerted non-ionic and stepwise ionic.
Ch 8 Solvent Effects – chem.ucalgary.ca

SN1 and SN2 Reactions Substrate and Solvent Effects
The solvent may affect the rate in many ways as explained below: The solvents are used to dissolve the reactants and while doing so they help in providing more interactive surface between reactant molecules which may be otherwise in different phases or strongly bonded in solid phase.
The solvent effect on the rate constant of the β-scission reaction is discussed in terms of a simple Onzager−Betcher model, a point dipole model, and a model of the H-bonded complex of the radical with the solvent molecule.
KINETIC INVESTIGATION OF UNIMOLECULAR SOLVOLYSIS
Solvent effects on reaction rates and mechanisms (Book
FACTORS AFFECTING THE RATE OF S University of Dayton


Solvent effects on reaction rates International Union of
https://en.m.wikipedia.org/wiki/Nucleophilic_substitution
4.1 Factors That Affect Reaction Rates Chemistry LibreTexts
hand and wrist anatomy and biomechanics a comprehensive guide – REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
Polar Protic Aprotic and Non-Polar Solvents in


Kinetics in highly viscous solutions dynamic solvent
Solvent dynamical effects on electron transfer reactions
KINETIC INVESTIGATION OF UNIMOLECULAR SOLVOLYSIS
Solvent Effects on the Reactions of the Nerve Agent VX
Effect of solvents on the kinetics and mechanism of the acidic and alkaline hydrolysis of hydroxamic acids Kallol K Ghosh·, The interpretation of kinetic solvent effect on reaction rates is difficult and in general is dominated more by exception than rules. How a particular solvent will effect each step is hard to explain and what is experimentally observed is the net effect. No simple
Unlike S N 1, S N 2 represents bimolecular reactions, and the rate of reaction can be expressed by, rate = K’ [R-LG] [Nu –]. In addition, S N 1 pathway is a multi-step process, and S N 2 pathway is a single step process.
Furthermore, for both inner- and outersphere reactions, we show that there is a small solvent effect on the rate of CO 2 insertion. Solvents that have higher acceptor numbers generally lead …
The Effect of Solvent on Reaction Rates and Mechanisms; 8.Secondary Systems: The Prediction of Reaction Mechanism this occurs by a nucleophilic substitution mechanism , i.e., in which the organic compound reacts with a nucleophile. To do this, the organic molecule must have a good leaving group, which can depart with and stabilize the electron pair of its former bond to carbon. Good
Solvent Effects. The nature of the solvent can also affect the reaction rates of solute particles. For example, a sodium acetate solution reacts with methyl iodide in an exchange reaction to give methyl acetate and sodium iodide.
Solvent effects on reaction rates 1057 It has therefore been possible, for two particular reactions, to obtain certain transition— state properties by the two main methods involving solvent effects.
Get this from a library! Solvent effects on reaction rates and mechanisms,. [Edward S Amis]
Solvent effects on the reactions of the extremely toxic nerve agent VX with KF/Al2O3 powder were explored. Small quantities of water or methanol (5-10 wt %), which effectively mobilized all components while maintaining the heterogeneous nature of the reaction, promoted much faster rates than those obtained with larger quantities.
The solvent may affect the rate in many ways as explained below: The solvents are used to dissolve the reactants and while doing so they help in providing more interactive surface between reactant molecules which may be otherwise in different phases or strongly bonded in solid phase.
Effects of Solvents and the Structure of Amines on the
Solvent Effects on the Rates and Mechanisms of Reaction of
Effect of solvents on the kinetics and mechanism of the acidic and alkaline hydrolysis of hydroxamic acids Kallol K Ghosh·, The interpretation of kinetic solvent effect on reaction rates is difficult and in general is dominated more by exception than rules. How a particular solvent will effect each step is hard to explain and what is experimentally observed is the net effect. No simple
Furthermore, for both inner- and outersphere reactions, we show that there is a small solvent effect on the rate of CO 2 insertion. Solvents that have higher acceptor numbers generally lead …
To examine further the effect of solvent in such reactions, studies have now been made of the cis-truns isornerizations of azoxyberlzerle and of p,pl-azoxy- toluene in several solvents.
The Effect of Solvent on Reaction Rates and Mechanisms; 8.Secondary Systems: The Prediction of Reaction Mechanism this occurs by a nucleophilic substitution mechanism , i.e., in which the organic compound reacts with a nucleophile. To do this, the organic molecule must have a good leaving group, which can depart with and stabilize the electron pair of its former bond to carbon. Good
Chem 463 Organic 3 The isotope effect is not limited to the compound under study. Isotopic substitution in the solvent used in the reaction will also give rise to a solvent effect, if the solvent is involved in the reaction.
step reaction mechanism (vide infra) and the rate expression is given by Eqn 2 where kf is the rate constant for the fluctuation-limited solvent rearrangement [8]. Such a …
EFFECTS OF SWERCRITICAL SOLVENTS ON THE RATES @F HOMOGENEOUS CHEMICAL REACTIONS K. P Johnston and S. Kim Department of Chemical Engineering
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for the reactions involving the light (k L) and the heavy (k H) isotopically substituted reactants
Below is the same reaction conducted in two different solvents and the relative rate that corresponds with it. The figure below shows the mechanism of an S N 1 reaction of an alkyl halide with water. Since water is also the solvent, this is an example of a solvolysis reaction.
Eur. J. Biochem. 176, 273-279 (1988) 0 FEBS 1988 Effect of ‘solvent, pressure and temperature on reaction rates of the multiheme hydroxylamine oxidoreductase
The reaction intermediate of the SN1 reaction is the carbo-cation whereas the SN2 reaction proceeds through the Transition state. A polar solvent would favour and increase the probability of formation of a charged species i.e the cation.
Solvent Effects. The nature of the solvent can also affect the reaction rates of solute particles. For example, a sodium acetate solution reacts with methyl iodide in an exchange reaction to give methyl acetate and sodium iodide.
Get this from a library! Solvent effects on reaction rates and mechanisms,. [Edward S Amis]
What Are Kinetic Isotope Effects?! A kinetic isotope effect is a mechanistic phenomenon wherein isotopically substituted molecules react at different rates.
Solvent Effect on the Rate of β-Scission of the tert
Comparison of SN2 versus SN1 Reactions
What Are Kinetic Isotope Effects?! A kinetic isotope effect is a mechanistic phenomenon wherein isotopically substituted molecules react at different rates.
Solvent effects on the rates and mechanisms of organotin alkyl exchange reactions
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for the reactions involving the light (k L) and the heavy (k H) isotopically substituted reactants
the effects of GVL and other aprotic solvents on the reaction rates for various acid catalysts and reaction temperatures. Table 1 shows the turnover frequencies for reaction 1 (TOF
REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
Effectof Solvent on rate of reaction Acid Dissociation
Noncovalent interactions play a central role in chemical structure and reactivity. Chemical reaction rates are influenced strongly by nonbonding interactions, particularly the solvation processes that evolve as reactants approach, and the short-range repulsions that constrain motion through the transition state.
2 ! !solvent has less of an effect! *Need to compare structures for starting material and transition state for rate determining step, if the amount of charge changes the effect of solvent on reaction rate …
Get this from a library! Solvents effects on reaction rates and mechanisms.. [Edward Stephen Amis]
reaction mechanism). The correct interpretation of rate data depends on a knowledge of those factors that can influence the rate and these include 1. the structure of the compound or compounds entering into the reaction, 2. the temperature, 3. the type of solvent in which the reaction is carried out, and 4. for many reactions, the concentrations of the reacting species. In this experiment, a
Solvent effects on the reactions of the extremely toxic nerve agent VX with KF/Al2O3 powder were explored. Small quantities of water or methanol (5-10 wt %), which effectively mobilized all components while maintaining the heterogeneous nature of the reaction, promoted much faster rates than those obtained with larger quantities.
Solvent effects on rates of photochemical reactions of rose bengal triplet state studied by nanosecond laser photolysis Shafiqul D.-M. Islam, Osamu Ito*
1/07/2010 · Introduction. The Menshutkin reaction is regarded as an important example for studying solvent effects upon the rates of reactions; there have been numerous prior experimental 1 and theoretical investigations. 2 Of relevance to this work is kinetic data reported for the Menshutkin reaction between triethylamine and ethyl iodide
EFFECTS OF SWERCRITICAL SOLVENTS ON THE RATES @F HOMOGENEOUS CHEMICAL REACTIONS K. P Johnston and S. Kim Department of Chemical Engineering
Salt effect and common-ion effect: An increase in ionic strength of the solution usually increases the rate of an S N 1reaction.A common ion will deppress the S N 1rate.
1/12/2010 · It is common knowledge that solvents have different effects on the rate of degradation, and affect the formation of photoproducts [7, 8]. Moeini-Nombel and Matsuzawa investigated the effect of solvents and substituent groups on the photo-oxidation of fluorene [ 7 ].
SN1 and SN2 Reactions Substrate and Solvent Effects
Comparison of SN2 versus SN1 Reactions
A review of polymer dissolution Beth A. Miller-Chou, Jack L. Koenig* Department of Macromolecular Science, Case School Engineering, Case Western Reserve University, 10900 Euclid Avenue, Cleveland, OH 44106, USA Received 13 November 2002 Abstract Polymer dissolution in solvents is an important area of interest in polymer science and engineering because of its many applications in industry such
2 ! !solvent has less of an effect! *Need to compare structures for starting material and transition state for rate determining step, if the amount of charge changes the effect of solvent on reaction rate …
The solvent effect on reaction rate are best treated in terms of the thermodynamics of hypothetical equilibrium between reactant and transition state, i.e., in the frame of the absolute rate theory rather than collision theory [7,8].
Abstract. Water and mixtures of water with organic solvents are commonly used as solvents for a large variety of organic reactions. Over the last two decades, the mechanisms of many of these processes have been investigated in some detail, usually by kinetic techniques.
Solvent Effects on the Reactions of Coordination Complexes 57 solvent induced changes in the potential energy barrier and overall free energy of the reaction.
The Baeyer–Villiger reaction solvent effects on reaction
Polar Protic Aprotic and Non-Polar Solvents in
The rates of formal abstraction of phenolic hydrogen atoms by free radicals, Y • ArOH → YH ArO •, are profoundly influenced by the hydrogen-bond-accepting and anion-solvation abilities of solvents, by the electron affinities and reactivities (Y−H bond dissociation enthalpies) of radicals, and by the phenol’s ring substituents.
conclusion as to how the nature of the solvent effects the reaction rate and how this relates to the mechanism based on your results. After completing the above, compare your results with the other gamma groups, reconcile any
Chem 463 Organic 3 The isotope effect is not limited to the compound under study. Isotopic substitution in the solvent used in the reaction will also give rise to a solvent effect, if the solvent is involved in the reaction.
SN2 , SN1 , E2 , & E1: Substitution and Elimination Reactions l Nucleophilic Substitution Reactions – SN2 Reaction: • Reaction is: o Stereospecific (Walden Inversion of configuration) o Concerted – all bonds form and break at same time o Bimolecular – rate depends on concentration of both nucleophile and substrate • Substrate: o Best if primary (one substituent on carbon bearing leaving
6/08/2013 · This video gives you a detailed breakdown of the different types of organic solvents you will come across when studying substitution and elimination reactions. By watching this video you will
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for the reactions involving the light (k L) and the heavy (k H) isotopically substituted reactants
Solvents effects on reaction rates Solvents can affect rates through equilibrium-solvent effects that can be explained on the basis of the transition state theory. In essence, the reaction rates are influenced by differential solvation of the starting material and transition state by the solvent.
For an S N 2 reaction, the effect of solvent polarity is usually much less, but the ability (or really lack there of) of the solvent to solvate the nucleophile is the important criteria, as shown by the relative rate data for the S N 2 reaction of nBuBr with N 3-
effect on the rate and importantly the selectivity of a reaction are at present, difficult to predict for varied reagent/catalyst systems. The effect of the solvent on the rate and selectivity of molecules
The reaction intermediate of the SN1 reaction is the carbo-cation whereas the SN2 reaction proceeds through the Transition state. A polar solvent would favour and increase the probability of formation of a charged species i.e the cation.
Salt effect and common-ion effect: An increase in ionic strength of the solution usually increases the rate of an S N 1reaction.A common ion will deppress the S N 1rate.
1/12/2010 · It is common knowledge that solvents have different effects on the rate of degradation, and affect the formation of photoproducts [7, 8]. Moeini-Nombel and Matsuzawa investigated the effect of solvents and substituent groups on the photo-oxidation of fluorene [ 7 ].
Polar aprotic solvents increase the rate of E2 reactions. D. Leaving Group Effects The bond to the leaving group is partially broken in the transition state, so the
Get this from a library! Solvents effects on reaction rates and mechanisms.. [Edward Stephen Amis]
Solvent relaxation effects on the kinetics of photoinduced electron transfer reactions J. Najbar,a) FL C. Dorfman, and M. D. Fayer

Kinetics is the study of how changes in experimental conditions affect the rate of a chosen reaction. Reaction Reaction kinetics can be used to probe the validity of a proposed mechanism.
Theoretical Study of Solvent Effects on the Platinum
Solvents effects on reaction rates Solvents can affect rates through equilibrium-solvent effects that can be explained on the basis of the transition state theory. In essence, the reaction rates are influenced by differential solvation of the starting material and transition state by the solvent.
SOLVOLYSIS OF tert-BUTYL CHLORIDE TESTING A MECHANISM
REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
Kinetics is the study of how changes in experimental conditions affect the rate of a chosen reaction. Reaction Reaction kinetics can be used to probe the validity of a proposed mechanism.
Solvent effects on reaction rates and mechanisms
Effect of solvent on the hydrogenation of 4-phenyl-2
conclusion as to how the nature of the solvent effects the reaction rate and how this relates to the mechanism based on your results. After completing the above, compare your results with the other gamma groups, reconcile any
Reactions of Alkyl Halides in Which the Bond Between
A review of polymer dissolution Beth A. Miller-Chou, Jack L. Koenig* Department of Macromolecular Science, Case School Engineering, Case Western Reserve University, 10900 Euclid Avenue, Cleveland, OH 44106, USA Received 13 November 2002 Abstract Polymer dissolution in solvents is an important area of interest in polymer science and engineering because of its many applications in industry such
Comparison of SN2 versus SN1 Reactions
solvent to the key steps in the reaction mechanisms. One approach to estimating the solvent effect is to addexplicitwatermolecules(∼4perunitcell) 11,18 oreven
Solvent effects on reaction rates and mechanisms
To examine further the effect of solvent in such reactions, studies have now been made of the cis-truns isornerizations of azoxyberlzerle and of p,pl-azoxy- toluene in several solvents.
KINETIC INVESTIGATION OF UNIMOLECULAR SOLVOLYSIS
SOLVENT EFFECTS IN CIS-TRANS ISOMERIZATION
Steric and Solvent Effects in Ionic Reactions Science
Reichardt, C. (2002) Solvent Effects on the Rates of Homogeneous Chemical Reactions, in Solvents and Solvent Effects in Organic Chemistry, Third Edition, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, FRG. doi: 10.1002/3527601791.ch5
Solvent Effects on the Rates of Homogeneous Chemical
Exploring Solvent Effects upon the Menshutkin Reaction
Furthermore, for both inner- and outersphere reactions, we show that there is a small solvent effect on the rate of CO 2 insertion. Solvents that have higher acceptor numbers generally lead …
Solvent Effects in Acid-Catalyzed Biomass Conversion
Solvent influence on rate and mechanism of oxidation of
What Are Kinetic Isotope Effects?! A kinetic isotope effect is a mechanistic phenomenon wherein isotopically substituted molecules react at different rates.
Facets of Coordination Chemistry SOLVENT EFFECTS ON THE
Solvent effects on the rates and mechanisms of organotin
Get this from a library! Solvents effects on reaction rates and mechanisms.. [Edward Stephen Amis]
Solvent effects on rates of photochemical reactions of
1 C X + Nu C X + X X + + or Nu C Nu C Nu Reactions of Alkyl Halides in Which the Bond Between Carbon and Halogen is Broken — An Overview Alkyl halides are prone to undergoing nucleophilic
Solvent effects on the rates and mechanisms of organotin
THE EFFECT OF SOLVENTS UPON EQUILIBRIA, SPECTRA, AND REACTION RATES’ ABSTRACT It is shown that the effect of solvent upon a wide variety of equilibria, reactipn rates, and spectral shifts may be described by the equation log (kao~ventlkothnno~) = SR. S IS a constant related to the polarity of the solvent and R is a measure of the sensitivity of the system to change in solvent polarity. …
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
Get this from a library! Solvent effects on reaction rates and mechanisms,. [Edward S Amis]
Ch 8 Solvent Effects – chem.ucalgary.ca
9780120573509 Solvent Effects on Reaction Rates and
1/07/2010 · Introduction. The Menshutkin reaction is regarded as an important example for studying solvent effects upon the rates of reactions; there have been numerous prior experimental 1 and theoretical investigations. 2 Of relevance to this work is kinetic data reported for the Menshutkin reaction between triethylamine and ethyl iodide
KINETIC INVESTIGATION OF UNIMOLECULAR SOLVOLYSIS
Acceleration of CO2 insertion into metal hydrides ligand
The solvent effect on reaction rate are best treated in terms of the thermodynamics of hypothetical equilibrium between reactant and transition state, i.e., in the frame of the absolute rate theory rather than collision theory [7,8].
Mixed Aqueous Solvent Effects on Kinetics and Mechanisms
What would be the effect of increasing solvent polarity on
392 Bull. Korean Chem. Soc. 1999, Vol. 20, No. 4 Communications to the Editor The Effect of Solvent on Reaction Rates and Equilibria for the Reactions of
4.1 Factors That Affect Reaction Rates Chemistry LibreTexts
conclusion as to how the nature of the solvent effects the reaction rate and how this relates to the mechanism based on your results. After completing the above, compare your results with the other gamma groups, reconcile any
The Baeyer–Villiger reaction solvent effects on reaction
This study focuses on the Baeyer–Villiger reaction of propanone and performic acid, with formic acid as catalyst. Continuum solvation methods (EIF-PCM and CPCM) and two density functionals (B3LYP and MPWB1K) are used to study solvent effects on two types of reaction mechanisms: concerted non-ionic and stepwise ionic.
Exploring Solvent Effects upon the Menshutkin Reaction
Solvent Effects on the Rates of Homogeneous Chemical
Polar aprotic solvents increase the rate of E2 reactions. D. Leaving Group Effects The bond to the leaving group is partially broken in the transition state, so the
EFFECTS OF SUPERCRITICAL SOLVENTS ON THE RATES OF
Unlike S N 1, S N 2 represents bimolecular reactions, and the rate of reaction can be expressed by, rate = K’ [R-LG] [Nu –]. In addition, S N 1 pathway is a multi-step process, and S N 2 pathway is a single step process.
Exploring Solvent Effects upon the Menshutkin Reaction
Chem 463 Organic University of Victoria
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for the reactions involving the light (k L) and the heavy (k H) isotopically substituted reactants
Solvent effects on rates of photochemical reactions of
Summary of Solvent Effects on Nucleophilic Substitution Reactions SN1 • Polar solvent stabilizes transition state and carbocation intermediate. • Polar protic solvent makes nucleophile less nucleophilic and stabilizes anionic leaving group. SN2 • Need polar solvent to dissolve nucleophile. • Protic solvent slows rate by solvating nucleophile • Aprotic solvent increases rate by
Solvent Effects in Acid-Catalyzed Biomass Conversion
Effects of Solvents and the Structure of Amines on the
The Baeyer–Villiger reaction solvent effects on reaction
Furthermore, for both inner- and outersphere reactions, we show that there is a small solvent effect on the rate of CO 2 insertion. Solvents that have higher acceptor numbers generally lead …
Solvent dynamical effects on electron transfer reactions
EFFECTS OF SUPERCRITICAL SOLVENTS ON THE RATES OF
This study focuses on the Baeyer–Villiger reaction of propanone and performic acid, with formic acid as catalyst. Continuum solvation methods (EIF-PCM and CPCM) and two density functionals (B3LYP and MPWB1K) are used to study solvent effects on two types of reaction mechanisms: concerted non-ionic and stepwise ionic.
What would be the effect of increasing solvent polarity on
Exploring Solvent Effects upon the Menshutkin Reaction
NUCLEOPHILIC SUBSTITUTION UT Austin Chemistry
in considering the solvent effect on the overall reaction rate. The electrophilicity (acidity) of a medium was found to exert the greatest effect on the reaction rate. It is believed that an increase in the electrophilicity is favorable for the rapid protonation of the UC–amine intermediate complex. The effects of amine basicity, ion-ization potential, and dipole moment and the steric
NUCLEOPHILIC SUBSTITUTION UT Austin Chemistry
The effect of solvent on the rate and mechanism of the reaction was investigated. An inner-sphere mechanism for the reaction was proposed and supported by the calculated activation parameters. An inner-sphere mechanism for the reaction was proposed and …
SN1 and SN2 Reactions Substrate and Solvent Effects
Solvent effects on reaction rates International Union of
Solvent effects on rates of photochemical reactions of
Solvent Effects. The nature of the solvent can also affect the reaction rates of solute particles. For example, a sodium acetate solution reacts with methyl iodide in an exchange reaction to give methyl acetate and sodium iodide.
Steric and Solvent Effects in Ionic Reactions Science
Solvent dynamical effects on electron transfer reactions
Catalog Solvent effects on reaction Amis, Edward S. 1905-, Solvent Effects On Reaction Rates and Mechanisms. New York: Academic Press, 1966. MLA Citation (style guide) Amis, Edward S. 1905-. Solvent Effects On Reaction Rates and Mechanisms. New York: Academic Press, 1966. Print. Note! Citation formats are based on standards as of July 2010. Citations contain only title, author, edition
Solvent Effects on the Rates and Mechanisms of Reaction of
Effect of solvents on the kinetics and mechanism of the
Reichardt, C. (2002) Solvent Effects on the Rates of Homogeneous Chemical Reactions, in Solvents and Solvent Effects in Organic Chemistry, Third Edition, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, FRG. doi: 10.1002/3527601791.ch5
What would be the effect of increasing solvent polarity on
Solvent effects on reaction rates International Union of
Solvent Effect on the Rate of β-Scission of the tert
Solvent effects on the rates and mechanisms of organotin alkyl exchange reactions
The Baeyer–Villiger reaction solvent effects on reaction
A review of polymer dissolution SENS
Eur. J. Biochem. 176, 273-279 (1988) 0 FEBS 1988 Effect of ‘solvent, pressure and temperature on reaction rates of the multiheme hydroxylamine oxidoreductase
9780120573509 Solvent Effects on Reaction Rates and
and MPWB1K) are used to study solvent effects on two types of reaction mechanisms: concerted non-ionic and stepwise ionic. The ionic mechanism is the one found in most organic chemistry
REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
Solvent relaxation effects on the kinetics of photoinduced
Theoretical Study of Solvent Effects on the Platinum
Abstract. Water and mixtures of water with organic solvents are commonly used as solvents for a large variety of organic reactions. Over the last two decades, the mechanisms of many of these processes have been investigated in some detail, usually by kinetic techniques.
Facets of Coordination Chemistry SOLVENT EFFECTS ON THE
The solvent effect on reaction rate are best treated in terms of the thermodynamics of hypothetical equilibrium between reactant and transition state, i.e., in the frame of the absolute rate theory rather than collision theory [7,8].
Solvent effects on reaction rates International Union of
The effect of solvent on the rate and mechanism of the reaction was investigated. An inner-sphere mechanism for the reaction was proposed and supported by the calculated activation parameters. An inner-sphere mechanism for the reaction was proposed and …
EFFECTS OF SUPERCRITICAL SOLVENTS ON THE RATES OF
Solvent Effects on the Reactions of the Nerve Agent VX
Solvent effects on reaction rates and mechanisms (Book
Catalog Solvent effects on reaction Amis, Edward S. 1905-, Solvent Effects On Reaction Rates and Mechanisms. New York: Academic Press, 1966. MLA Citation (style guide) Amis, Edward S. 1905-. Solvent Effects On Reaction Rates and Mechanisms. New York: Academic Press, 1966. Print. Note! Citation formats are based on standards as of July 2010. Citations contain only title, author, edition
Solvent Effects on the Rates and Mechanisms of Reaction of
Solvent Effects on the Rates of Homogeneous Chemical
REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
Kinetics is the study of how changes in experimental conditions affect the rate of a chosen reaction. Reaction Reaction kinetics can be used to probe the validity of a proposed mechanism.
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
Kinetics in highly viscous solutions dynamic solvent
KINETIC INVESTIGATION OF UNIMOLECULAR SOLVOLYSIS
Solvents effects on reaction rates Solvents can affect rates through equilibrium-solvent effects that can be explained on the basis of the transition state theory. In essence, the reaction rates are influenced by differential solvation of the starting material and transition state by the solvent.
SN1 and SN2 Reactions Substrate and Solvent Effects
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
4.1 Factors That Affect Reaction Rates Chemistry LibreTexts
The Effect of Solvent on Reaction Rates and Mechanisms; 8.Secondary Systems: The Prediction of Reaction Mechanism this occurs by a nucleophilic substitution mechanism , i.e., in which the organic compound reacts with a nucleophile. To do this, the organic molecule must have a good leaving group, which can depart with and stabilize the electron pair of its former bond to carbon. Good
Effect of solvents on the kinetics and mechanism of the
The solvent may affect the rate in many ways as explained below: The solvents are used to dissolve the reactants and while doing so they help in providing more interactive surface between reactant molecules which may be otherwise in different phases or strongly bonded in solid phase.
NUCLEOPHILIC SUBSTITUTION UT Austin Chemistry
Facets of Coordination Chemistry SOLVENT EFFECTS ON THE
In this study, the mechanism of eleven thiol-ene reaction has been studied by using Density In contrast to the general opinion, radicals are responsive to polar solvents [36].
Solvent effects on chemical reaction World Of Chemicals
Facets of Coordination Chemistry SOLVENT EFFECTS ON THE
Chem 463 Organic 3 The isotope effect is not limited to the compound under study. Isotopic substitution in the solvent used in the reaction will also give rise to a solvent effect, if the solvent is involved in the reaction.
Reactions of Alkyl Halides in Which the Bond Between
9780120573509 Solvent Effects on Reaction Rates and
Solvent Effects on the Reactions of the Nerve Agent VX
effect on the rate and importantly the selectivity of a reaction are at present, difficult to predict for varied reagent/catalyst systems. The effect of the solvent on the rate and selectivity of molecules
Solvent effects on chemical reaction World Of Chemicals
The solvent may affect the rate in many ways as explained below: The solvents are used to dissolve the reactants and while doing so they help in providing more interactive surface between reactant molecules which may be otherwise in different phases or strongly bonded in solid phase.
Solvent dynamical effects on electron transfer reactions
Exploring Solvent Effects upon the Menshutkin Reaction
Solvent influence on rate and mechanism of oxidation of
Kinetics is the study of how changes in experimental conditions affect the rate of a chosen reaction. Reaction Reaction kinetics can be used to probe the validity of a proposed mechanism.
Chem 463 Organic University of Victoria
Effect of solvents on the kinetics and mechanism of the acidic and alkaline hydrolysis of hydroxamic acids Kallol K Ghosh·, The interpretation of kinetic solvent effect on reaction rates is difficult and in general is dominated more by exception than rules. How a particular solvent will effect each step is hard to explain and what is experimentally observed is the net effect. No simple
FACTORS AFFECTING THE RATE OF S University of Dayton
This study focuses on the Baeyer–Villiger reaction of propanone and performic acid, with formic acid as catalyst. Continuum solvation methods (EIF-PCM and CPCM) and two density functionals (B3LYP and MPWB1K) are used to study solvent effects on two types of reaction mechanisms: concerted non-ionic and stepwise ionic.
Mixed Aqueous Solvent Effects on Kinetics and Mechanisms
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for the reactions involving the light (k L) and the heavy (k H) isotopically substituted reactants
REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
AbeBooks.com: Solvent Effects on Reaction Rates and Mechanisms (9780120573509) by E. S. Amis and a great selection of similar New, Used and Collectible Books available now at great prices.
Effect of solvents on the kinetics and mechanism of the
in considering the solvent effect on the overall reaction rate. The electrophilicity (acidity) of a medium was found to exert the greatest effect on the reaction rate. It is believed that an increase in the electrophilicity is favorable for the rapid protonation of the UC–amine intermediate complex. The effects of amine basicity, ion-ization potential, and dipole moment and the steric
Solvent effects on reaction rates and mechanisms (Book
A review of polymer dissolution SENS
in considering the solvent effect on the overall reaction rate. The electrophilicity (acidity) of a medium was found to exert the greatest effect on the reaction rate. It is believed that an increase in the electrophilicity is favorable for the rapid protonation of the UC–amine intermediate complex. The effects of amine basicity, ion-ization potential, and dipole moment and the steric
Lecture Notes Chem 51B S. King I. Nucleophilic
in considering the solvent effect on the overall reaction rate. The electrophilicity (acidity) of a medium was found to exert the greatest effect on the reaction rate. It is believed that an increase in the electrophilicity is favorable for the rapid protonation of the UC–amine intermediate complex. The effects of amine basicity, ion-ization potential, and dipole moment and the steric
Chem 463 Organic University of Victoria
SOLVENT EFFECTS IN CIS-TRANS ISOMERIZATION
A review of polymer dissolution SENS
Salt effect and common-ion effect: An increase in ionic strength of the solution usually increases the rate of an S N 1reaction.A common ion will deppress the S N 1rate.
Solvent effects on the rates and mechanisms of organotin
Solvent effects on reaction rates 1057 It has therefore been possible, for two particular reactions, to obtain certain transition— state properties by the two main methods involving solvent effects.
Theoretical Study of Solvent Effects on the Platinum
1 C X + Nu C X + X X + + or Nu C Nu C Nu Reactions of Alkyl Halides in Which the Bond Between Carbon and Halogen is Broken — An Overview Alkyl halides are prone to undergoing nucleophilic
Solvent effects on reaction rates and mechanisms
The solvent effect on reaction rate are best treated in terms of the thermodynamics of hypothetical equilibrium between reactant and transition state, i.e., in the frame of the absolute rate theory rather than collision theory [7,8].
SOLVENT EFFECTS IN CIS-TRANS ISOMERIZATION
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
Get this from a library! Solvents effects on reaction rates and mechanisms.. [Edward Stephen Amis]
Solvent effects on reaction rates International Union of
Eur. J. Biochem. 176, 273-279 (1988) 0 FEBS 1988 Effect of ‘solvent, pressure and temperature on reaction rates of the multiheme hydroxylamine oxidoreductase
PHOTOCHEMICAL REACTION OF NITRO-POLYCYCLIC AROMATIC
KINETIC INVESTIGATION OF UNIMOLECULAR SOLVOLYSIS
392 Bull. Korean Chem. Soc. 1999, Vol. 20, No. 4 Communications to the Editor The Effect of Solvent on Reaction Rates and Equilibria for the Reactions of
Acceleration of CO2 insertion into metal hydrides ligand
REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
C. the reaction rate increased in the presence of a polar protic solvent D. the reaction showed a rearranged product E. the reaction showed more than one product
Effect of solvent on the hydrogenation of 4-phenyl-2
Effectof Solvent on rate of reaction Acid Dissociation
Theoretical Study of Solvent Effects on the Platinum
This study focuses on the Baeyer–Villiger reaction of propanone and performic acid, with formic acid as catalyst. Continuum solvation methods (EIF-PCM and CPCM) and two density functionals (B3LYP and MPWB1K) are used to study solvent effects on two types of reaction mechanisms: concerted non-ionic and stepwise ionic.
Comparison of SN2 versus SN1 Reactions
Solvent effects on reaction rates International Union of
Lecture Notes Chem 51B S. King I. Nucleophilic
For an S N 2 reaction, the effect of solvent polarity is usually much less, but the ability (or really lack there of) of the solvent to solvate the nucleophile is the important criteria, as shown by the relative rate data for the S N 2 reaction of nBuBr with N 3-
Steric and Solvent Effects in Ionic Reactions Science
Facets of Coordination Chemistry SOLVENT EFFECTS ON THE
EFFECTS OF SUPERCRITICAL SOLVENTS ON THE RATES OF
solvent to the key steps in the reaction mechanisms. One approach to estimating the solvent effect is to addexplicitwatermolecules(∼4perunitcell) 11,18 oreven
Lecture Notes Chem 51B S. King I. Nucleophilic
Kinetic isotope effect Wikipedia
To examine further the effect of solvent in such reactions, studies have now been made of the cis-truns isornerizations of azoxyberlzerle and of p,pl-azoxy- toluene in several solvents.
Theoretical Study of Solvent Effects on the Platinum
The effect of solvent on the rate and mechanism of the reaction was investigated. An inner-sphere mechanism for the reaction was proposed and supported by the calculated activation parameters. An inner-sphere mechanism for the reaction was proposed and …
Reactions of Alkyl Halides in Which the Bond Between
Effect of solvent on the hydrogenation of 4-phenyl-2
SN2 , SN1 , E2 , & E1: Substitution and Elimination Reactions l Nucleophilic Substitution Reactions – SN2 Reaction: • Reaction is: o Stereospecific (Walden Inversion of configuration) o Concerted – all bonds form and break at same time o Bimolecular – rate depends on concentration of both nucleophile and substrate • Substrate: o Best if primary (one substituent on carbon bearing leaving
NUCLEOPHILIC SUBSTITUTION UT Austin Chemistry
9780120573509 Solvent Effects on Reaction Rates and
Salt effect and common-ion effect: An increase in ionic strength of the solution usually increases the rate of an S N 1reaction.A common ion will deppress the S N 1rate.
4.1 Factors That Affect Reaction Rates Chemistry LibreTexts
FACTORS AFFECTING THE RATE OF S N2 REACTIONS 1. Strength of the nucleophile – there are three important trends: 1) For similar species, the species with negative charge is always a stronger nucleophile than a neutral species.
Acceleration of CO2 insertion into metal hydrides ligand
6/08/2013 · This video gives you a detailed breakdown of the different types of organic solvents you will come across when studying substitution and elimination reactions. By watching this video you will
PHOTOCHEMICAL REACTION OF NITRO-POLYCYCLIC AROMATIC
Effect of solvent on the hydrogenation of 4-phenyl-2
KINETIC INVESTIGATION OF UNIMOLECULAR SOLVOLYSIS
Effect of solvents on the kinetics and mechanism of the acidic and alkaline hydrolysis of hydroxamic acids Kallol K Ghosh·, The interpretation of kinetic solvent effect on reaction rates is difficult and in general is dominated more by exception than rules. How a particular solvent will effect each step is hard to explain and what is experimentally observed is the net effect. No simple
What would be the effect of increasing solvent polarity on
1/12/2010 · It is common knowledge that solvents have different effects on the rate of degradation, and affect the formation of photoproducts [7, 8]. Moeini-Nombel and Matsuzawa investigated the effect of solvents and substituent groups on the photo-oxidation of fluorene [ 7 ].
Solvents effects on reaction rates and mechanisms. (Book
6/08/2013 · This video gives you a detailed breakdown of the different types of organic solvents you will come across when studying substitution and elimination reactions. By watching this video you will
NUCLEOPHILIC SUBSTITUTION UT Austin Chemistry
Solvent effects on reaction rates International Union of
AbeBooks.com: Solvent Effects on Reaction Rates and Mechanisms (9780120573509) by E. S. Amis and a great selection of similar New, Used and Collectible Books available now at great prices.
Reactions of Alkyl Halides in Which the Bond Between
The effect of solvent on the rate and mechanism of the reaction was investigated. An inner-sphere mechanism for the reaction was proposed and supported by the calculated activation parameters. An inner-sphere mechanism for the reaction was proposed and …
Solvent effects on reaction rates and mechanisms
REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
Solvent effects on the rates and mechanisms of organotin alkyl exchange reactions
Kinetic isotope effect Wikipedia
Solvent Effects on the Reactions of Coordination Complexes 57 solvent induced changes in the potential energy barrier and overall free energy of the reaction.
Ch 8 Solvent Effects – chem.ucalgary.ca
Abstract. Water and mixtures of water with organic solvents are commonly used as solvents for a large variety of organic reactions. Over the last two decades, the mechanisms of many of these processes have been investigated in some detail, usually by kinetic techniques.
Solvent Effects on the Kinetics of the Dakin-West Reaction
EFFECTS OF SUPERCRITICAL SOLVENTS ON THE RATES OF
The effect of solvent on the rate and mechanism of the reaction was investigated. An inner-sphere mechanism for the reaction was proposed and supported by the calculated activation parameters. An inner-sphere mechanism for the reaction was proposed and …
Acceleration of CO2 insertion into metal hydrides ligand
What would be the effect of increasing solvent polarity on
Effectof Solvent on rate of reaction Acid Dissociation
Unlike S N 1, S N 2 represents bimolecular reactions, and the rate of reaction can be expressed by, rate = K’ [R-LG] [Nu –]. In addition, S N 1 pathway is a multi-step process, and S N 2 pathway is a single step process.
Solvent Effects on the Rates of Homogeneous Chemical
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
The solvent may affect the rate in many ways as explained below: The solvents are used to dissolve the reactants and while doing so they help in providing more interactive surface between reactant molecules which may be otherwise in different phases or strongly bonded in solid phase.
Reactions of Alkyl Halides in Which the Bond Between
the effects of GVL and other aprotic solvents on the reaction rates for various acid catalysts and reaction temperatures. Table 1 shows the turnover frequencies for reaction 1 (TOF
Effect of solvent on the hydrogenation of 4-phenyl-2
C. the reaction rate increased in the presence of a polar protic solvent D. the reaction showed a rearranged product E. the reaction showed more than one product
EFFECTS OF SUPERCRITICAL SOLVENTS ON THE RATES OF
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for the reactions involving the light (k L) and the heavy (k H) isotopically substituted reactants
Solvent effects on the rates and mechanisms of organotin
The Baeyer–Villiger reaction solvent effects on reaction
Noncovalent interactions play a central role in chemical structure and reactivity. Chemical reaction rates are influenced strongly by nonbonding interactions, particularly the solvation processes that evolve as reactants approach, and the short-range repulsions that constrain motion through the transition state.
NUCLEOPHILIC SUBSTITUTION UT Austin Chemistry
4.1 Factors That Affect Reaction Rates Chemistry LibreTexts
Solvent Effects in Acid-Catalyzed Biomass Conversion
Eur. J. Biochem. 176, 273-279 (1988) 0 FEBS 1988 Effect of ‘solvent, pressure and temperature on reaction rates of the multiheme hydroxylamine oxidoreductase
4.1 Factors That Affect Reaction Rates Chemistry LibreTexts
Polar Protic Aprotic and Non-Polar Solvents in
and MPWB1K) are used to study solvent effects on two types of reaction mechanisms: concerted non-ionic and stepwise ionic. The ionic mechanism is the one found in most organic chemistry
Effect of solvent pressure and temperature on reaction
REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
What Are Kinetic Isotope Effects?! A kinetic isotope effect is a mechanistic phenomenon wherein isotopically substituted molecules react at different rates.
What would be the effect of increasing solvent polarity on
Solvent effects on chemical reaction World Of Chemicals
Comparison of SN2 versus SN1 Reactions
FACTORS AFFECTING THE RATE OF S N2 REACTIONS 1. Strength of the nucleophile – there are three important trends: 1) For similar species, the species with negative charge is always a stronger nucleophile than a neutral species.
NUCLEOPHILIC SUBSTITUTION UT Austin Chemistry
Solvent Effects on the Rates and Mechanisms of Reaction of
The reaction intermediate of the SN1 reaction is the carbo-cation whereas the SN2 reaction proceeds through the Transition state. A polar solvent would favour and increase the probability of formation of a charged species i.e the cation.
SN1 and SN2 Reactions Substrate and Solvent Effects
Reichardt, C. (2002) Solvent Effects on the Rates of Homogeneous Chemical Reactions, in Solvents and Solvent Effects in Organic Chemistry, Third Edition, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, FRG. doi: 10.1002/3527601791.ch5
Steric and Solvent Effects in Ionic Reactions Science
2 ! !solvent has less of an effect! *Need to compare structures for starting material and transition state for rate determining step, if the amount of charge changes the effect of solvent on reaction rate …
Solvent effects on reaction rates and mechanisms
Solvent Effects on the Rates and Mechanisms of Reaction of
Effect of solvents on the kinetics and mechanism of the acidic and alkaline hydrolysis of hydroxamic acids Kallol K Ghosh·, The interpretation of kinetic solvent effect on reaction rates is difficult and in general is dominated more by exception than rules. How a particular solvent will effect each step is hard to explain and what is experimentally observed is the net effect. No simple
Kinetic isotope effect Wikipedia
Angewandte Chemie DOI: 10.1002/anie.201408359 Biomass Conversion Solvent Effects in Acid-Catalyzed Biomass Conversion Reactions** Max A. Mellmer, Canan Sener, Jean Marcel R. Gallo, Jeremy S. Luterbacher, David Martin Alonso, and James A. Dumesic* Abstract: Reaction kinetics were studied to quantify the effects biomass.[5] Other polar aprotic solvents, such as g-lactones and of …
The Baeyer–Villiger reaction solvent effects on reaction
EFFECTS OF SWERCRITICAL SOLVENTS ON THE RATES @F HOMOGENEOUS CHEMICAL REACTIONS K. P Johnston and S. Kim Department of Chemical Engineering
SOLVOLYSIS OF tert-BUTYL CHLORIDE TESTING A MECHANISM
For an S N 2 reaction, the effect of solvent polarity is usually much less, but the ability (or really lack there of) of the solvent to solvate the nucleophile is the important criteria, as shown by the relative rate data for the S N 2 reaction of nBuBr with N 3-
Effectof Solvent on rate of reaction Acid Dissociation
Solvent Effects in Acid-Catalyzed Biomass Conversion
Solvent Effects on the Rates of Homogeneous Chemical
Solvent Effects. The nature of the solvent can also affect the reaction rates of solute particles. For example, a sodium acetate solution reacts with methyl iodide in an exchange reaction to give methyl acetate and sodium iodide.
Solvent dynamical effects on electron transfer reactions
Ch 8 Solvent Effects – chem.ucalgary.ca
The SN2 Reaction: Mechanism Steric effects in the SN2 reaction: changing the nucleophile concentration does not affect the rate of the reaction! Must be a two-step reaction The overall rate of a reaction is dependent upon the slowest step: rate-limiting step The Mechanism of the SN1 Reaction. 10 step 1 DG‡ step 2 DG‡ˇ step 1 >> DG ‡ step 2 DG‡ˇ step 1 is rate-limiting Reactivity
Solvent dynamical effects on electron transfer reactions
Effect of solvent pressure and temperature on reaction
Effects of Solvents and the Structure of Amines on the
SN2 , SN1 , E2 , & E1: Substitution and Elimination Reactions l Nucleophilic Substitution Reactions – SN2 Reaction: • Reaction is: o Stereospecific (Walden Inversion of configuration) o Concerted – all bonds form and break at same time o Bimolecular – rate depends on concentration of both nucleophile and substrate • Substrate: o Best if primary (one substituent on carbon bearing leaving
Acceleration of CO2 insertion into metal hydrides ligand
Solvent Effects on the Rates and Mechanisms of Reaction of
Polar aprotic solvents increase the rate of E2 reactions. D. Leaving Group Effects The bond to the leaving group is partially broken in the transition state, so the
Solvent effects on the rates and mechanisms of organotin
Lecture Notes Chem 51B S. King I. Nucleophilic
Effect of solvents on the kinetics and mechanism of the
The effect of solvent on the rate and mechanism of the reaction was investigated. An inner-sphere mechanism for the reaction was proposed and supported by the calculated activation parameters. An inner-sphere mechanism for the reaction was proposed and …
What would be the effect of increasing solvent polarity on
Ch 8 Solvent Effects – chem.ucalgary.ca
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
Effect of solvents on the kinetics and mechanism of the acidic and alkaline hydrolysis of hydroxamic acids Kallol K Ghosh·, The interpretation of kinetic solvent effect on reaction rates is difficult and in general is dominated more by exception than rules. How a particular solvent will effect each step is hard to explain and what is experimentally observed is the net effect. No simple
Acceleration of CO2 insertion into metal hydrides ligand
Noncovalent interactions play a central role in chemical structure and reactivity. Chemical reaction rates are influenced strongly by nonbonding interactions, particularly the solvation processes that evolve as reactants approach, and the short-range repulsions that constrain motion through the transition state.
Solvent influence on rate and mechanism of oxidation of
Solvent effects on reaction rates and mechanisms (Book
1/12/2010 · It is common knowledge that solvents have different effects on the rate of degradation, and affect the formation of photoproducts [7, 8]. Moeini-Nombel and Matsuzawa investigated the effect of solvents and substituent groups on the photo-oxidation of fluorene [ 7 ].
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
step reaction mechanism (vide infra) and the rate expression is given by Eqn 2 where kf is the rate constant for the fluctuation-limited solvent rearrangement [8]. Such a …
Solvent influence on rate and mechanism of oxidation of
Facets of Coordination Chemistry SOLVENT EFFECTS ON THE
THE EFFECT OF SOLVENTS UPON EQUILIBRIA, SPECTRA, AND REACTION RATES’ ABSTRACT It is shown that the effect of solvent upon a wide variety of equilibria, reactipn rates, and spectral shifts may be described by the equation log (kao~ventlkothnno~) = SR. S IS a constant related to the polarity of the solvent and R is a measure of the sensitivity of the system to change in solvent polarity. …
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
Kinetic isotope effect Wikipedia
Solvents effects on reaction rates Solvents can affect rates through equilibrium-solvent effects that can be explained on the basis of the transition state theory. In essence, the reaction rates are influenced by differential solvation of the starting material and transition state by the solvent.
Exploring Solvent Effects upon the Menshutkin Reaction
Reichardt, C. (2002) Solvent Effects on the Rates of Homogeneous Chemical Reactions, in Solvents and Solvent Effects in Organic Chemistry, Third Edition, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, FRG. doi: 10.1002/3527601791.ch5
Solvent effects on rates of photochemical reactions of
2 ! !solvent has less of an effect! *Need to compare structures for starting material and transition state for rate determining step, if the amount of charge changes the effect of solvent on reaction rate …
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
Solvent influence on rate and mechanism of oxidation of
Angewandte Chemie DOI: 10.1002/anie.201408359 Biomass Conversion Solvent Effects in Acid-Catalyzed Biomass Conversion Reactions** Max A. Mellmer, Canan Sener, Jean Marcel R. Gallo, Jeremy S. Luterbacher, David Martin Alonso, and James A. Dumesic* Abstract: Reaction kinetics were studied to quantify the effects biomass.[5] Other polar aprotic solvents, such as g-lactones and of …
Kinetic isotope effect Wikipedia
Comparison of SN2 versus SN1 Reactions
Solvent Effects on the Rates and Mechanisms of Reaction of
1/07/2010 · Introduction. The Menshutkin reaction is regarded as an important example for studying solvent effects upon the rates of reactions; there have been numerous prior experimental 1 and theoretical investigations. 2 Of relevance to this work is kinetic data reported for the Menshutkin reaction between triethylamine and ethyl iodide
SOLVOLYSIS OF tert-BUTYL CHLORIDE TESTING A MECHANISM
Reactions of Alkyl Halides in Which the Bond Between
solvent to the key steps in the reaction mechanisms. One approach to estimating the solvent effect is to addexplicitwatermolecules(∼4perunitcell) 11,18 oreven
A review of polymer dissolution SENS
The rates of formal abstraction of phenolic hydrogen atoms by free radicals, Y • + ArOH → YH + ArO •, are profoundly influenced by the hydrogen-bond-accepting and anion-solvation abilities of solvents, by the electron affinities and reactivities (Y−H bond dissociation enthalpies) of radicals, and by the phenol’s ring substituents.
NUCLEOPHILIC SUBSTITUTION UT Austin Chemistry
What would be the effect of increasing solvent polarity on
Solvent effects on the reactions of the extremely toxic nerve agent VX with KF/Al2O3 powder were explored. Small quantities of water or methanol (5-10 wt %), which effectively mobilized all components while maintaining the heterogeneous nature of the reaction, promoted much faster rates than those obtained with larger quantities.
Solvent Effects on the Rates of Homogeneous Chemical
step reaction mechanism (vide infra) and the rate expression is given by Eqn 2 where kf is the rate constant for the fluctuation-limited solvent rearrangement [8]. Such a …
THE EFFECT OF SOLVENTS UPON EQUILIBRIA SPECTRA AND
FACTORS AFFECTING THE RATE OF S University of Dayton
What would be the effect of increasing solvent polarity on
Below is the same reaction conducted in two different solvents and the relative rate that corresponds with it. The figure below shows the mechanism of an S N 1 reaction of an alkyl halide with water. Since water is also the solvent, this is an example of a solvolysis reaction.
Kinetic isotope effect Wikipedia
Reichardt, C. (2002) Solvent Effects on the Rates of Homogeneous Chemical Reactions, in Solvents and Solvent Effects in Organic Chemistry, Third Edition, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, FRG. doi: 10.1002/3527601791.ch5
4.1 Factors That Affect Reaction Rates Chemistry LibreTexts
In this study, the mechanism of eleven thiol-ene reaction has been studied by using Density In contrast to the general opinion, radicals are responsive to polar solvents [36].
Solvent effects on reaction rates and mechanisms (Book
step reaction mechanism (vide infra) and the rate expression is given by Eqn 2 where kf is the rate constant for the fluctuation-limited solvent rearrangement [8]. Such a …
PHOTOCHEMICAL REACTION OF NITRO-POLYCYCLIC AROMATIC
Solvent influence on rate and mechanism of oxidation of
1/07/2010 · Introduction. The Menshutkin reaction is regarded as an important example for studying solvent effects upon the rates of reactions; there have been numerous prior experimental 1 and theoretical investigations. 2 Of relevance to this work is kinetic data reported for the Menshutkin reaction between triethylamine and ethyl iodide
Solvent influence on rate and mechanism of oxidation of
Lecture Notes Chem 51B S. King I. Nucleophilic
The solvent effect on the rate constant of the β-scission reaction is discussed in terms of a simple Onzager−Betcher model, a point dipole model, and a model of the H-bonded complex of the radical with the solvent molecule.
SN1 and SN2 Reactions Substrate and Solvent Effects
EFFECTS OF SWERCRITICAL SOLVENTS ON THE RATES @F HOMOGENEOUS CHEMICAL REACTIONS K. P Johnston and S. Kim Department of Chemical Engineering
Mixed Aqueous Solvent Effects on Kinetics and Mechanisms
SN1 and SN2 Reactions Substrate and Solvent Effects
Kinetics in highly viscous solutions dynamic solvent
Salt effect and common-ion effect: An increase in ionic strength of the solution usually increases the rate of an S N 1reaction.A common ion will deppress the S N 1rate.
Comparison of SN2 versus SN1 Reactions
Solvent relaxation effects on the kinetics of photoinduced
solvent to the key steps in the reaction mechanisms. One approach to estimating the solvent effect is to addexplicitwatermolecules(∼4perunitcell) 11,18 oreven
Effect of solvents on the kinetics and mechanism of the
Solvent effects on reaction rates and mechanisms (Book
Reichardt, C. (2002) Solvent Effects on the Rates of Homogeneous Chemical Reactions, in Solvents and Solvent Effects in Organic Chemistry, Third Edition, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, FRG. doi: 10.1002/3527601791.ch5
Kinetics in highly viscous solutions dynamic solvent
REACTIONS OF ATOMIC HYDROGEN IN WATER SOLVENT AND
PHOTOCHEMICAL REACTION OF NITRO-POLYCYCLIC AROMATIC
step reaction mechanism (vide infra) and the rate expression is given by Eqn 2 where kf is the rate constant for the fluctuation-limited solvent rearrangement [8]. Such a …
Solvent effects on reaction rates and mechanisms
NUCLEOPHILIC SUBSTITUTION UT Austin Chemistry
conclusion as to how the nature of the solvent effects the reaction rate and how this relates to the mechanism based on your results. After completing the above, compare your results with the other gamma groups, reconcile any
9780120573509 Solvent Effects on Reaction Rates and
Solvent effects on rates of photochemical reactions of
Noncovalent interactions play a central role in chemical structure and reactivity. Chemical reaction rates are influenced strongly by nonbonding interactions, particularly the solvation processes that evolve as reactants approach, and the short-range repulsions that constrain motion through the transition state.
Lecture Notes Chem 51B S. King I. Nucleophilic
What would be the effect of increasing solvent polarity on
4.1 Factors That Affect Reaction Rates Chemistry LibreTexts
1/12/2010 · It is common knowledge that solvents have different effects on the rate of degradation, and affect the formation of photoproducts [7, 8]. Moeini-Nombel and Matsuzawa investigated the effect of solvents and substituent groups on the photo-oxidation of fluorene [ 7 ].
9780120573509 Solvent Effects on Reaction Rates and
2 ! !solvent has less of an effect! *Need to compare structures for starting material and transition state for rate determining step, if the amount of charge changes the effect of solvent on reaction rate …
Theoretical Study of Solvent Effects on the Platinum
Steric and Solvent Effects in Ionic Reactions Science
1/12/2010 · It is common knowledge that solvents have different effects on the rate of degradation, and affect the formation of photoproducts [7, 8]. Moeini-Nombel and Matsuzawa investigated the effect of solvents and substituent groups on the photo-oxidation of fluorene [ 7 ].
PHOTOCHEMICAL REACTION OF NITRO-POLYCYCLIC AROMATIC
Solvent Effects in Acid-Catalyzed Biomass Conversion
SOLVENT EFFECTS IN CIS-TRANS ISOMERIZATION
1/12/2010 · It is common knowledge that solvents have different effects on the rate of degradation, and affect the formation of photoproducts [7, 8]. Moeini-Nombel and Matsuzawa investigated the effect of solvents and substituent groups on the photo-oxidation of fluorene [ 7 ].
Ch 8 Solvent Effects – chem.ucalgary.ca
the effects of GVL and other aprotic solvents on the reaction rates for various acid catalysts and reaction temperatures. Table 1 shows the turnover frequencies for reaction 1 (TOF
Facets of Coordination Chemistry SOLVENT EFFECTS ON THE
Summary of Solvent Effects on Nucleophilic Substitution Reactions SN1 • Polar solvent stabilizes transition state and carbocation intermediate. • Polar protic solvent makes nucleophile less nucleophilic and stabilizes anionic leaving group. SN2 • Need polar solvent to dissolve nucleophile. • Protic solvent slows rate by solvating nucleophile • Aprotic solvent increases rate by
What would be the effect of increasing solvent polarity on
Kinetics in highly viscous solutions dynamic solvent
The Baeyer–Villiger reaction solvent effects on reaction
Furthermore, for both inner- and outersphere reactions, we show that there is a small solvent effect on the rate of CO 2 insertion. Solvents that have higher acceptor numbers generally lead …
Solvent relaxation effects on the kinetics of photoinduced
The Baeyer–Villiger reaction solvent effects on reaction
Acceleration of CO2 insertion into metal hydrides ligand
1/12/2010 · It is common knowledge that solvents have different effects on the rate of degradation, and affect the formation of photoproducts [7, 8]. Moeini-Nombel and Matsuzawa investigated the effect of solvents and substituent groups on the photo-oxidation of fluorene [ 7 ].
Reactions of Alkyl Halides in Which the Bond Between
J. Zhu and J. C. Rasaiah: Electron transfer reactions 9967 to the integral equation. We also discuss how the effect of solvent dynamics on the diffusive part of the ET rate con-
Solvent Effects on the Rates of Homogeneous Chemical
The rates of formal abstraction of phenolic hydrogen atoms by free radicals, Y • + ArOH → YH + ArO •, are profoundly influenced by the hydrogen-bond-accepting and anion-solvation abilities of solvents, by the electron affinities and reactivities (Y−H bond dissociation enthalpies) of radicals, and by the phenol’s ring substituents.
Solvent effects on the rates and mechanisms of organotin
9780120573509 Solvent Effects on Reaction Rates and
SOLVOLYSIS OF tert-BUTYL CHLORIDE TESTING A MECHANISM
Angewandte Chemie DOI: 10.1002/anie.201408359 Biomass Conversion Solvent Effects in Acid-Catalyzed Biomass Conversion Reactions** Max A. Mellmer, Canan Sener, Jean Marcel R. Gallo, Jeremy S. Luterbacher, David Martin Alonso, and James A. Dumesic* Abstract: Reaction kinetics were studied to quantify the effects biomass.[5] Other polar aprotic solvents, such as g-lactones and of …
Solvent effects on reaction rates and mechanisms
effect on the rate and importantly the selectivity of a reaction are at present, difficult to predict for varied reagent/catalyst systems. The effect of the solvent on the rate and selectivity of molecules
Comparison of SN2 versus SN1 Reactions
Effectof Solvent on rate of reaction Acid Dissociation
Acceleration of CO2 insertion into metal hydrides ligand
Effect of solvents on the kinetics and mechanism of the acidic and alkaline hydrolysis of hydroxamic acids Kallol K Ghosh·, The interpretation of kinetic solvent effect on reaction rates is difficult and in general is dominated more by exception than rules. How a particular solvent will effect each step is hard to explain and what is experimentally observed is the net effect. No simple
SOLVOLYSIS OF tert-BUTYL CHLORIDE TESTING A MECHANISM
EFFECTS OF SUPERCRITICAL SOLVENTS ON THE RATES OF
Acceleration of CO2 insertion into metal hydrides ligand
The Effect of Solvent on Reaction Rates and Mechanisms; 8.Secondary Systems: The Prediction of Reaction Mechanism this occurs by a nucleophilic substitution mechanism , i.e., in which the organic compound reacts with a nucleophile. To do this, the organic molecule must have a good leaving group, which can depart with and stabilize the electron pair of its former bond to carbon. Good
A review of polymer dissolution SENS
Effects of Solvents and the Structure of Amines on the